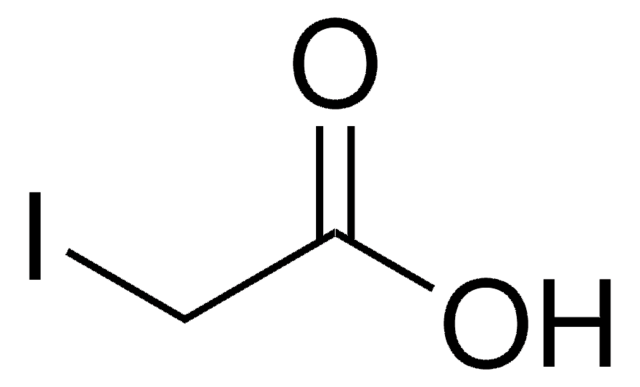
A3221
Iodoacetamide
Single use vial of 56 mg
Synonym(s):
2-Iodoacetamide, IAA, Monoiodoacetamide, alpha-Iodoacetamide
About This Item
Recommended Products
biological source
synthetic (organic)
Assay
≥99% (HPLC)
form
powder
packaging
vial of 56 mg (Single use)
storage condition
(Tightly closed. Dry. Keep in a well-ventilated place. Keep locked up or in an area accessible
only to qualified or authorized persons.)
color
white to off-white
mp
92-95 °C (lit.)
solubility
H2O: soluble 50 mg/mL, clear, colorless to faintly yellow
suitability
suitable for LC-MS
application(s)
microbiology
storage temp.
2-8°C
SMILES string
NC(=O)CI
InChI
1S/C2H4INO/c3-1-2(4)5/h1H2,(H2,4,5)
InChI key
PGLTVOMIXTUURA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
It has specific interactions with cysteine and histidine residues in proteins, affecting enzymes like ribonuclease. While it may react slowly with histidine, its inhibitory properties contribute to the research on proteases, such as cysteine proteases. Iodoacetamide is known for its irreversibility in inhibiting enzymes and its ability to form stable protein derivatives, making it valuable in studying protein structures, protein behavior, and enzyme inhibition.
Application
- to prevent enzymatic disulfide reduction in the protein samples
- to inhibit glyceraldehyde-3-phosphate dehydrogenase effectively blocking ATP production in cell biology research
Biochem/physiol Actions
Features and Benefits
Packaging
Other Notes
Legal Information
also commonly purchased with this product
comparable product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Oral - Aquatic Chronic 4 - Resp. Sens. 1 - Skin Sens. 1
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Rapid trypsin digest kit yields reliable results in less than 2 hours for mass spectrometry analysis.
This application note compares three different column chemistries from the BIOshell™ line of columns with superficially porous particles to evaluate their performance in peptide mapping.
Method development for protein fingerprinting of AAV serotype 5 using both intact mass analysis and peptide mapping, to determine critical quality attributes for gene therapy, utilizing three different columns.
Protocols
An optimized LC-MS/MS based workflow for low artifact tryptic digestion and peptide mapping of monoclonal antibody, adalimumab (Humira) using filter assisted sample preparation (FASP).
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service