900421
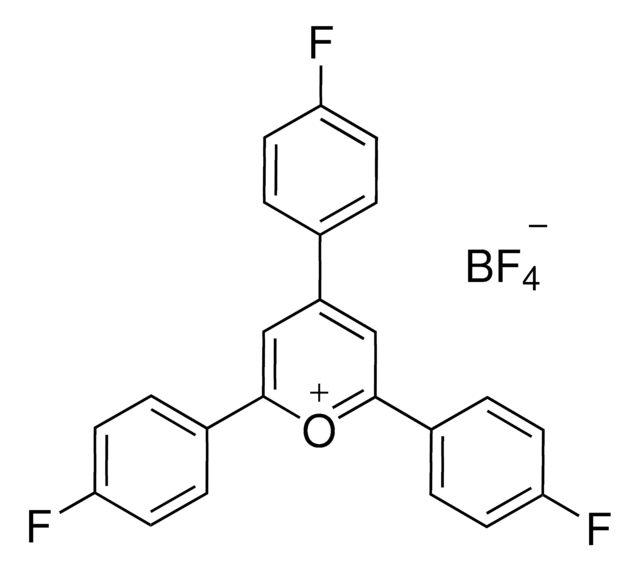
9-Mesityl-3,6-di-tert-butyl-10-phenylacridinium tetrafluoroborate
≥95%
Synonym(s):
3,6-Bis(1,1-dimethylethyl)-10-phenyl-9-(2,4,6-trimethylphenyl)-acridinium tetrafluoroborate, 3,6-Di-tert-butyl-9-(2,4,6-trimethylphenyl)-10-phenylacridinium tetrafluoroborate
About This Item
Recommended Products
Quality Level
Assay
≥95%
form
powder or crystals
reaction suitability
core: acridinium
reagent type: catalyst
reaction type: Photocatalysis
mp
277.2 °C
photocatalyst activation
455 nm
Related Categories
Application
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
related product
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
The Nicewicz lab is focused on the discovery of new and powerful reaction methodologies that proceed via the intermediacy of highly reactive cation radical species. Included in these transformations are anti-Markovnikov selective additions of amines, alcohols, carboxylic acids, amides and mineral acids to alkenes.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service





![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)
![[Ir(dtbbpy)(ppy)2]PF6](/deepweb/assets/sigmaaldrich/product/structures/158/329/2544d673-d267-4aa1-8f46-2652aad4bfa0/640/2544d673-d267-4aa1-8f46-2652aad4bfa0.png)
![[Ir(dFCF3ppy)2-(5,5’-dCF3bpy)]PF6 ≥95%](/deepweb/assets/sigmaaldrich/product/structures/422/901/e00f3148-fb86-4f94-9e79-21d064c3f327/640/e00f3148-fb86-4f94-9e79-21d064c3f327.png)
![Tris[2-phenylpyridinato-C2,N]iridium(III) 97%](/deepweb/assets/sigmaaldrich/product/structures/167/234/658d0b76-d31d-4fd5-8041-e04e207227c9/640/658d0b76-d31d-4fd5-8041-e04e207227c9.png)

![10-Ethyl-3,7,8-trimethyl-benzo[g]pteridine-2,4(3H,10H)-dione ≥95%](/deepweb/assets/sigmaaldrich/product/structures/610/004/664b92ed-0ce1-4e66-9f3e-d1cc53f1bc9a/640/664b92ed-0ce1-4e66-9f3e-d1cc53f1bc9a.png)