38907
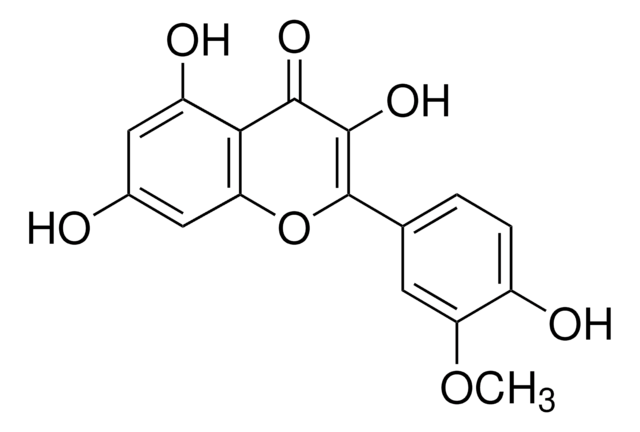
Isorhamnetin
analytical standard
Synonym(s):
3′-Methoxy-3,4′,5,7-tetrahydroxyflavone, 3′-Methylquercetin, 3,4′,5,7-Tetrahydroxy 3′-methoxyflavone, Isorhamnetol, Quercetin 3′-methyl ether
About This Item
Recommended Products
grade
analytical standard
Quality Level
Assay
≥99% (HPLC)
shelf life
limited shelf life, expiry date on the label
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
application(s)
food and beverages
format
neat
storage temp.
2-8°C
SMILES string
COc1cc(ccc1O)C2=C(O)C(=O)c3c(O)cc(O)cc3O2
InChI
1S/C16H12O7/c1-22-11-4-7(2-3-9(11)18)16-15(21)14(20)13-10(19)5-8(17)6-12(13)23-16/h2-6,17-19,21H,1H3
InChI key
IZQSVPBOUDKVDZ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- Rat plasma samples by ultra-high performance liquid chromatography-tandem mass spectrometry (UHPLC-MS/MS) equipped with electrospray ionization (ESI) and multiple reaction monitoring (MRM) mode of detection.
- Lotus (Nelumbo nucifera) leaves by macroporous resin chromatography combined with HPLC-ESI-MS/MS.
- Powdered extracts and finished products samples of Ginkgo biloba by HPLC-MS.
- Human plasma and urine by high-throughput micro solid-phase extraction (μ-SPE), followed by analysis using UHPLC in interface with quadrupole time-of-flight mass spectrometry (Q TOFMS).
Biochem/physiol Actions
Packaging
Other Notes
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service