D167800
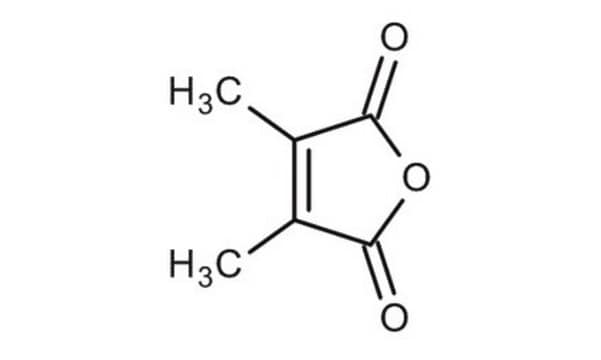
2,3-Dimethylmaleic anhydride
98%
Synonym(s):
Dimethylmaleic anhydride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C6H6O3
CAS Number:
Molecular Weight:
126.11
Beilstein:
112044
EC Number:
MDL number:
UNSPSC Code:
12162002
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
Assay
98%
form
flakes
bp
223 °C (lit.)
mp
93-96 °C (lit.)
SMILES string
CC1=C(C)C(=O)OC1=O
InChI
1S/C6H6O3/c1-3-4(2)6(8)9-5(3)7/h1-2H3
InChI key
MFGALGYVFGDXIX-UHFFFAOYSA-N
Application
Reagent used in the synthesis of maleimides and as an amino group protecting agent for superoxide dismutase.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Se Rim Yoon et al.
Journal of biomedical materials research. Part A, 100(8), 2027-2033 (2012-05-15)
A charge-converting and pH-dependent nanocarrier was achieved by conjugating 2,3-dimethylmaleic anhydride (DMMA) to the amino group of an octadecyl grafted poly (2-hydroxyethyl aspartamide) (PHEA-g-C(18)-NH(2)) backbone, thereby forming a spherical micelle. PHEA, a poly(amino acid)s derivative, was derived from poly(succinimide), which
Kabrambam D Singh et al.
Scientific reports, 7(1), 12483-12483 (2017-10-04)
Evolution of resistance among insects to action of pesticides has led to the discovery of several insecticides (neonicotinoids and organophosphates) with new targets in insect nervous system. Present study evaluates the mode of inhibition of acetylchlonesterase (AChE), biochemical efficacy, and
S Tsunoda et al.
The Journal of pharmacology and experimental therapeutics, 290(1), 368-372 (1999-06-25)
We attempted to develop a novel method for the chemical modification of cytokines with synthetic polymers to increase in vivo therapeutic efficacy. A pH-reversible amino-protective reagent, dimethylmaleic anhydride (DMMAn), was used for polymer conjugation of tumor necrosis factor-alpha (TNF-alpha) with
Hyeong Sup Yu et al.
Pharmaceutics, 11(6) (2019-06-05)
As caterpillars detect the presence of predators and secrete poison, herein, we show an innovative and highly effective cancer therapeutic system using biocompatible chitosan nanofiber (CNf) installed with a pH-responsive motif that senses tumor extracellular pH, pHe, prior to delivering
C Hikita et al.
The Journal of biological chemistry, 274(25), 17671-17676 (1999-06-11)
When an intercalated epithelial cell line was seeded at low density and allowed to reach confluence, it located the anion exchanger band 3 in the apical membrane and an H+-ATPase in the basolateral membrane. The same clonal cells seeded at
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service