P6635
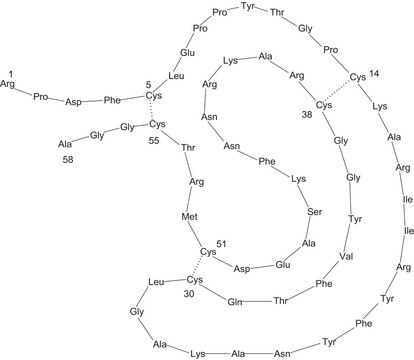
Phosphorylase b from rabbit muscle
lyophilized powder, ≥20 units/mg protein, 2× crystallization
Sinónimos:
α-Glucan Phosphorylase, 1,4-α-D-Glucan:orthophosphate α-D-glucosyltransferase, Glycogen Phosphorylase
About This Item
Productos recomendados
biological source
rabbit muscle
Quality Level
form
lyophilized powder
specific activity
≥20 units/mg protein
mol wt
97,200 Da by calculation
purified by
2× crystallization
storage condition
(Keep container tightly closed in a dry and well-ventilated place)
technique(s)
mass spectrometry (MS): suitable
impurities
~0.01 μmol/mg protein 5′-AMP (This low level will not interfere with phosphorylase and phosphorylase kinase assays.)
UniProt accession no.
foreign activity
phosphoglucomutase ≤1.0%
phosphorylase a ≤10%
phosphorylase kinase ≤0.5%
phosphorylase phosphatase, debrancher enzyme, AMPase and ATPase ≤0.1%
storage temp.
−20°C
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Glycogen phosphorylase (PG), a specialized complex allosteric enzyme has an evolutionarily conserved gene sequence. GP contains a family of three isozymes such as muscle GP (mGP), liver GP (lGP), and brain GP (bGP) in humans.
Application
- in the calibration of Sepharose C1-6B columns while studying the molecular weight of methylamine dehydrogenase subunits
- in ion mobility-mass spectrometry studies of phosphorylase B ions that have been generated with supercharging reagents, in the charge-reducing buffer
- for the preparation of p32 labeled phosphorylase A using phosphorylase kinase and [32P]ATP
- in phosphorylase phosphatase assay
- in enzyme assay as a positive control to ensure the reaction system for the activity determination was adopted
Biochem/physiol Actions
Packaging
Unit Definition
Physical form
antibody
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico