21906
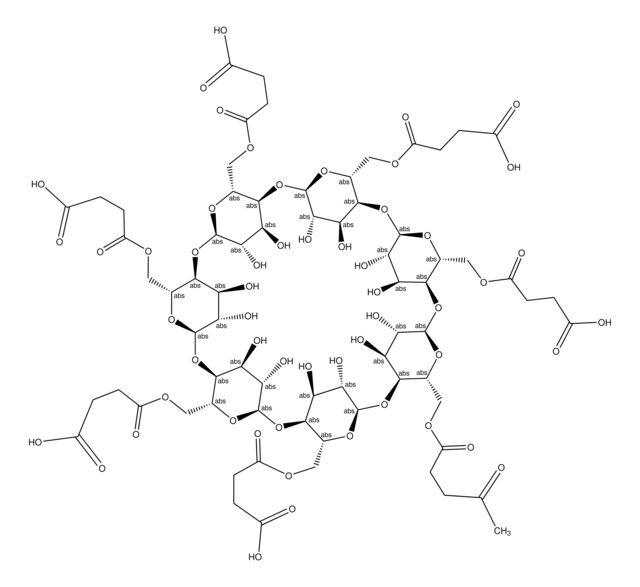
Carboxymethyl-β-cyclodextrin sodium salt
Sinónimos:
Sodium CM-β-Cyclodextrin
About This Item
Productos recomendados
form
solid
Quality Level
impurities
10% water
mp
~245 °C (dec.)
solubility
H2O: 50 mg/mL, clear to slightly hazy, colorless
SMILES string
O[C@H]1[C@H](O)[C@@H]2O[C@H]3O[C@H](COCC(O)=O)[C@@H](O[C@H]4O[C@H](COCC(O)=O)[C@@H](O[C@H]5O[C@H](COCC(O)=O)[C@@H](O[C@H]6O[C@H](COCC(O)=O)[C@@H](O[C@H]7O[C@H](COCC(O)=O)[C@@H](O[C@H]8O[C@H](COCC(O)=O)[C@@H](O[C@H]1O[C@@H]2COCC(O)=O)[C@@H](O)[C@@H]8O)[C@@H](O)[C@@H]7O)[C@@H](O)[C@@H]6O)[C@@H](O)[C@@H]5O)[C@@H](O)[C@@H]4O)[C@@H](O)[C@@H]3O
InChI
1S/C56H84O49/c57-22(58)8-85-1-15-43-29(71)36(78)50(92-15)100-44-16(2-86-9-23(59)60)94-52(38(80)31(44)73)102-46-18(4-88-11-25(63)64)96-54(40(82)33(46)75)104-48-20(6-90-13-27(67)68)98-56(42(84)35(48)77)105-49-21(7-91-14-28(69)70)97-55(41(83)34(49)76)103-47-19(5-89-12-26(65)66)95-53(39(81)32(47)74)101-45-17(3-87-10-24(61)62)93-51(99-43)37(79)30(45)72/h15-21,29-56,71-84H,1-14H2,(H,57,58)(H,59,60)(H,61,62)(H,63,64)(H,65,66)(H,67,68)(H,69,70)/t15-,16-,17-,18-,19-,20-,21-,29+,30+,31+,32+,33+,34+,35+,36+,37+,38+,39+,40+,41+,42+,43-,44-,45-,46-,47-,48-,49-,50-,51-,52-,53-,54-,55-,56-/m1/s1
InChI key
WROHVVIPQXODQM-LYSKQWNXSA-N
General description
Application
Carboxymethyl-β-cyclodextrin (CMBCD) is used in the development of recognition and separation technologies to resolve enantiomers based on chiral properties and size. Carboxymethyl-β-cyclodextrin is used in chiral selection and separation by capillary electrophoresis. Carboxymethyl-β-cyclodextrin is used in the development of drug delivery vehicles such as nanocarriers and as a nucleic acid transfection agent.
Analysis Note
Other Notes
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico