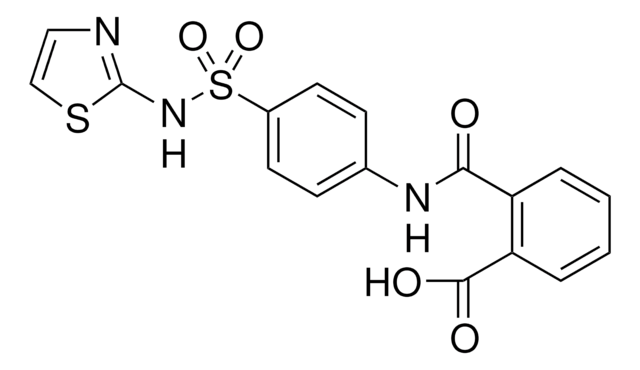
PHR1896
Exemestane Related Compound D
Pharmaceutical Secondary Standard; Certified Reference Material
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
UNSPSC Code:
41116107
NACRES:
NA.24
Productos recomendados
grade
certified reference material
pharmaceutical secondary standard
Quality Level
API family
exemestane
CofA
current certificate can be downloaded
packaging
pkg of 30 mg
application(s)
pharmaceutical
format
neat
storage temp.
2-8°C
General description
Exemestane Related Compound D is an impurity of the steroidal anticancer drug exemestane. Exemestane belongs to the class of antiestrogens known as aromatase inhibitors and it is commonly used for the treatment of breast cancer.
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Application
Exemestane may be used as a pharmaceutical reference standard for the determination of the analyte in bulk drug and pharmaceutical formulations by chromatography.
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
Analysis Note
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
Other Notes
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
Footnote
To see an example of a Certificate of Analysis for this material enter LRAB0340 in the Documents slot below. This is an example certificate only and may not be the lot that you receive.
related product
Referencia del producto
Descripción
Precios
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
Lot/Batch Number
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Analytical method validation for HPLC assay of oral anticancer drug exemestane
Yavuz B
Journal of Pharmaceutical Sciences, 32(1), 15-15 (2007)
A novel validated stability-indicating RP-HPLC method for the determination of Exemestane (steroidal aromatase inhibitor)
Mukthinuthalapati MA and Bukkapatnam V
Journal of Bioequivalence & Bioavailability, 7(6), 288-288 (2015)
Exemestane: a review of its clinical efficacy and safety
L?nning PE
Breast (Edinburgh, Scotland), 10(3), 198-208 (2001)
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico