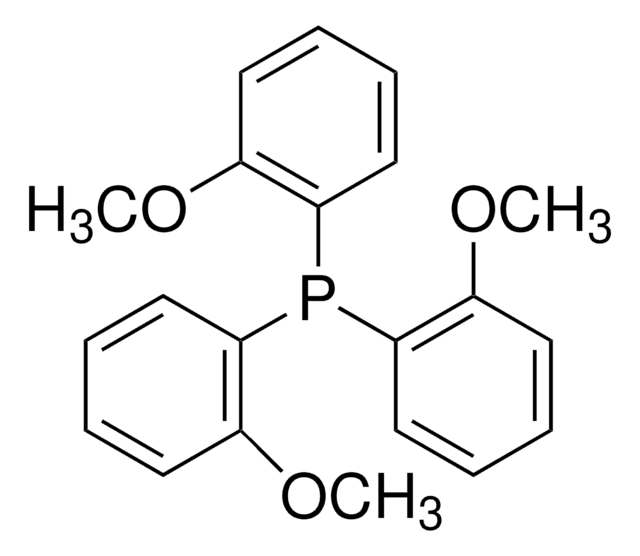
747270
4-(Diethylphosphino)-N,N-dimethylaniline
97%
Sinónimos:
4-(Diethylphosphino)-N,N-dimethylbenzenamine, Diethyl[4-(N,N-dimethylamino)phenyl]phosphine, Ellman Ligand
About This Item
Productos recomendados
assay
97%
form
liquid
reaction suitability
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: ligand
refractive index
n20/D 1.583 (lit.)
density
1.023 g/mL at 25 °C (lit.)
functional group
phosphine
storage temp.
2-8°C
SMILES string
CN(C)C1=CC=C(P(CC)CC)C=C1
InChI
1S/C12H20NP/c1-5-14(6-2)12-9-7-11(8-10-12)13(3)4/h7-10H,5-6H2,1-4H3
InChI key
FMNOWZMFKYVARU-UHFFFAOYSA-N
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Contenido relacionado
The Ellman group has participated in the development of a variety of C-H functionalization methods. An electron rich phosphine ligand has proven to be very useful for a variety of Rh(I)-catalyzed C-C bond forming reactions applicable to heterocycle synthesis as exemplified in the recent Science paper “Proton Donor Acidity Controls Selectivity in Nonaromatic Nitrogen Heterocycle Synthesis.” Another useful ligand developed for the highly functional group compatible direct arylation of nitrogen heterocycles is described in a 2008 J. Am. Chem. Soc. paper “Rh(I)-Catalyzed Arylation of Heterocycles via C-H Bond Activation: Expanded Scope through Mechanistic Insight.” The Ellman group also developed the chiral amine reagent tert-Butanesulfinamide, which is extensively used in academics and industry for the asymmetric synthesis of amines. A comprehensive survey of tert-Butanesulfinamide methods and applications up through 2009 is provided in the 2010 Chemical Reviews article, “Synthesis and Applications of tert-Butanesulfinamide.”
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico


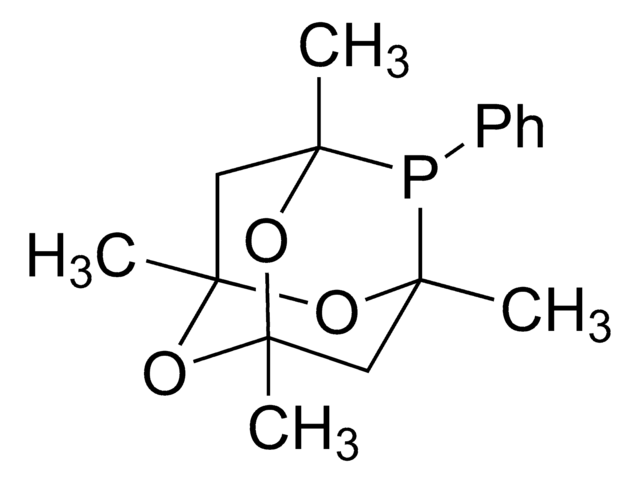

![5-[Di(1-adamantyl)phosphino]-1′,3′,5′-triphenyl-1′H-[1,4′]bipyrazole 97%](/deepweb/assets/sigmaaldrich/product/structures/334/020/2da77724-fc86-4d88-bc93-0b43ae777daa/640/2da77724-fc86-4d88-bc93-0b43ae777daa.png)