408395
Dimethylammonium dimethylcarbamate
Sinónimos:
N-Methylmethanamine dimethylcarbamate, Dimcarb, Dimethylamine carbon dioxide complex
About This Item
Productos recomendados
form
liquid
Quality Level
refractive index
n20/D 1.454 (lit.)
bp
60-61 °C (lit.)
density
1.05 g/mL at 25 °C (lit.)
functional group
amine
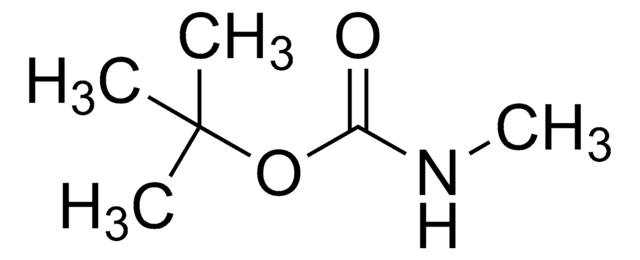
SMILES string
CNC.CN(C)C(O)=O
InChI
1S/C3H7NO2.C2H7N/c1-4(2)3(5)6;1-3-2/h1-2H3,(H,5,6);3H,1-2H3
InChI key
JIYXHCMRGZVYMA-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
Application
- For synthesis of calixarene-based ketocyanine fluorophores
- Distillation extraction of tannins from plant materials
- Electrodeposition of silver or of lead on glassy carbon and mercury film electrodes
- Synthesis of Ag and Au nanostructures
- Reusable reaction medium for synthesis of monoarylidene cyclopentanones
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Inhalation - Eye Dam. 1 - Flam. Liq. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
3 - Flammable liquids
wgk_germany
WGK 3
flash_point_f
55.4 °F - DIN 51758
flash_point_c
13 °C - DIN 51758
ppe
Eyeshields, Faceshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico