116238
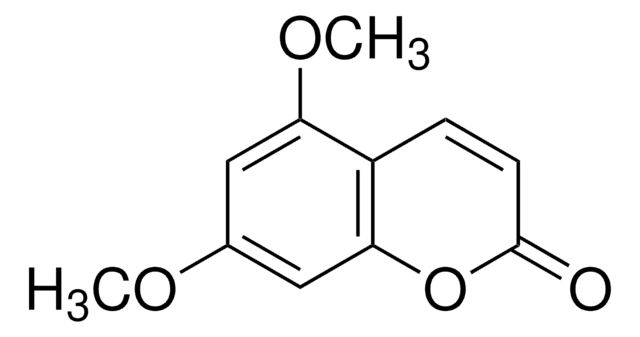
5,7-Dimethoxycoumarin
98%
Sinónimos:
Citropten, Limettin
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C11H10O4
Número de CAS:
Peso molecular:
206.19
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
assay
98%
form
solid
mp
146-149 °C (lit.)
functional group
ester
SMILES string
COc1cc(OC)c2C=CC(=O)Oc2c1
InChI
1S/C11H10O4/c1-13-7-5-9(14-2)8-3-4-11(12)15-10(8)6-7/h3-6H,1-2H3
InChI key
NXJCRELRQHZBQA-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
5,7-dimethoxycoumarin is isolated and identified from leaves and fruits of Pelea anisata H. Mann, a plant whose fruit are used in the construction of mohikana leis. It induces frameshift mutagenesis in bacteria. It also causes lethal photosensitization and the formation of sister chromatid exchanges in Chinese hamster cells.
Biochem/physiol Actions
5,7-Dimethoxycoumarin induces the processes of differentiation and melanogenesis in murine (B16) and human (A375).
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Daniela Alesiani et al.
International journal of oncology, 34(6), 1727-1735 (2009-05-09)
In this study, the processes of differentiation and melanogenesis induced by 5,7-dimethoxycoumarin in murine (B16) and human (A375) melanoma cells were investigated. Taking into account the previously demonstrated antiproliferative and differentiation activities of this compound, we examined Ras/Raf/Mek/Erk mitogen-activated protein
Takeshi Kinoshita et al.
Chemical & pharmaceutical bulletin, 50(1), 118-120 (2002-02-05)
A new C-8 prenylated 5,7-dimethoxycoumarin named omphamurrayin was isolated from the leaves of Murraya paniculata var. omphalocarpa, and its structure was established as 5,7-dimethoxy-8-(1-oxo-2-senecioyl-3-methyl-3-butenyl)-2H-1-benzopyran-2-one on the basis of the spectroscopic evidence. The taxonomic status of M. paniculata var. omphalocarpa is
S Makki et al.
Journal of chromatography, 563(2), 407-413 (1991-02-15)
Citropten (5,7-dimethoxycoumarin) and bergapten (5-methoxypsoralen) are present in bergamot oil which is used as a tanning cosmetic product. The aim of this study was to quantify, using high-performance liquid chromatography, the amount of citropten and bergapten in the skin after
V Jung et al.
Biochimica et biophysica acta, 740(1), 64-72 (1983-05-20)
The photobinding of 5,7-dimethoxycoumarin to isolated adenovirus-type 2 DNA has been investigated with respect to the influence of the ionic environment, and varying molar ratios of DNA(p): 5,7-dimethoxycoumarin. In particular, the ultraviolet radiation-induced covalent addition of 5,7-dimethoxycoumarin to adenovirus DNA
Isolation and characterization of the photoadducts of 5,7-dimethoxycoumarin and adenosine.
T H Cho et al.
Photochemistry and photobiology, 46(2), 305-309 (1987-08-01)
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico