All Photos(1)
About This Item
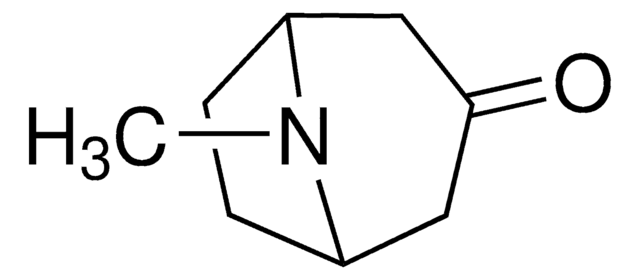
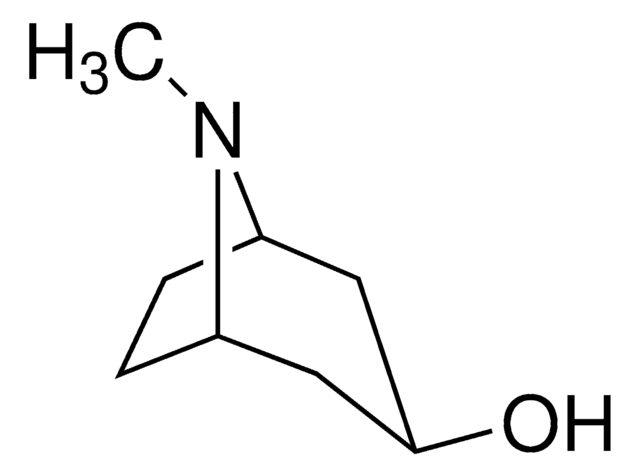
Empirical Formula (Hill Notation):
C8H15NO
CAS Number:
Molecular Weight:
141.21
Beilstein:
80188
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥97.0% (NT)
form
powder
impurities
0-3% water
solubility
H2O: 0.1 g/mL, clear
storage temp.
2-8°C
SMILES string
CN1[C@H]2CC[C@@H]1C[C@H](O)C2
InChI
1S/C8H15NO/c1-9-6-2-3-7(9)5-8(10)4-6/h6-8,10H,2-5H2,1H3/t6-,7+,8+
InChI key
CYHOMWAPJJPNMW-JIGDXULJSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Birgit Dräger
Phytochemistry, 67(4), 327-337 (2006-01-24)
Two stereospecific oxidoreductases constitute a branch point in tropane alkaloid metabolism. Products of tropane metabolism are the alkaloids hyoscyamine, scopolamine, cocaine, and polyhydroxylated nortropane alkaloids, the calystegines. Both tropinone reductases reduce the precursor tropinone to yield either tropine or pseudotropine.
Rawia Zayed et al.
Zeitschrift fur Naturforschung. C, Journal of biosciences, 59(11-12), 863-867 (2005-01-26)
Hairy root cultures of Brugmansia suaveolens were set up by infection of root tips with Agrobacterium rhizogenes. The successful transformation was confirmed by analysing rolC and virC genes using polymerase chain reaction (PCR). Hairy root cultures were employed to study
Laszlo Gyermek et al.
Journal of critical care, 24(1), 58-65 (2009-03-11)
There is a need for neuromuscular relaxant (NMR) agents that are of the "nondepolarizing type" and produce rapidly developing and short-lasting skeletal muscle relaxation in anesthesiology. Many efforts have been directed to produce such agents. Our research focused on the
Renata A Kwiecień et al.
Archives of biochemistry and biophysics, 510(1), 35-41 (2011-03-23)
(15)N heavy isotope effects are especially useful when detail is sought pertaining to the reaction mechanism for the cleavage of a C-N bond. Their potential in assisting to describe the mechanism of N-demethylation of tertiary amines by the action of
A Yamashita et al.
Biochemistry, 38(24), 7630-7637 (1999-07-01)
Tropinone reductase-II (TR-II) catalyzes the NADPH-dependent reduction of the carbonyl group of tropinone to a beta-hydroxyl group. The crystal structure of TR-II complexed with NADP+ and pseudotropine (psi-tropine) has been determined at 1.9 A resolution. A seven-residue peptide near the
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service