PHR1874
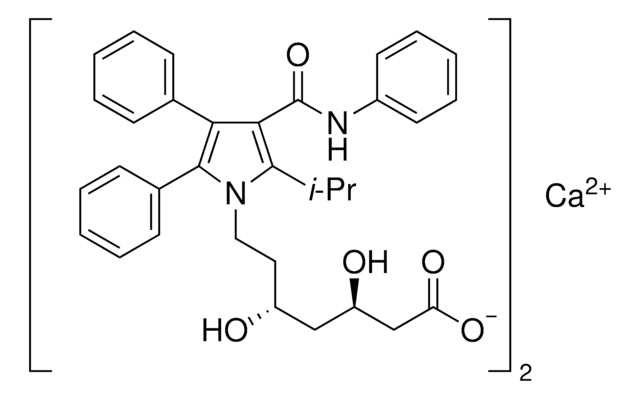
Atorvastatin Related Compound I
pharmaceutical secondary standard, certified reference material
Sinónimos:
tert-Butyl (4R,6R)-6-[2-[2-(4-fluorophenyl)-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)-1-pyrrolyl]ethyl]-2,2-dimethyl-1,3-dioxane-4-acetate, tert-Butyl 2-((4R,6R)-6-{2-[2-(4-fluorophenyl)-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)-1H-pyrrol-1-yl]ethyl}-2,2-dimethyl-1,3-dioxan-4-yl)acetate, (4R,6R)-6-[2-[2-(4-Fluorophenyl)-5-(1-methylethyl)-3-phenyl-4-[(phenylamino)carbonyl]-1H-pyrrol-1-yl]ethyl]-2,2-dimethyl-1,3-dioxane-4-acetic acid 1,1-dimethylethyl ester, tert-Butyl 2-[(4R,6R)-6-[2-[2-(4-fluorophenyl)-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)-1H-pyrrol-1-yl]ethyl]-2,2-dimethyl-1,3-dioxan-4-yl]acetate
About This Item
Productos recomendados
grade
certified reference material
pharmaceutical secondary standard
Quality Level
agency
traceable to USP 1044593
API family
atorvastatin
CofA
current certificate can be downloaded
packaging
pkg of 30 mg
mp
144-148 °C (lit.)
application(s)
pharmaceutical
format
neat
storage temp.
2-8°C
SMILES string
CC(C)c1c(C(=O)Nc2ccccc2)c(-c3ccccc3)c(-c4ccc(F)cc4)n1CC[C@@H]5C[C@H](CC(=O)OC(C)(C)C)OC(C)(C)O5
InChI
1S/C40H47FN2O5/c1-26(2)36-35(38(45)42-30-16-12-9-13-17-30)34(27-14-10-8-11-15-27)37(28-18-20-29(41)21-19-28)43(36)23-22-31-24-32(47-40(6,7)46-31)25-33(44)48-39(3,4)5/h8-21,26,31-32H,22-25H2,1-7H3,(H,42,45)/t31-,32-/m1/s1
InChI key
NPPZOMYSGNZDKY-ROJLCIKYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Application
Analysis Note
Other Notes
Footnote
related product
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico