53889
Bacoside A3
analytical standard
Sinónimos:
(3β,16β,23R)-16,23:16,30-Diepoxy-20-hydroxydammar-24-en-3-yl O-α-L-arabinofuranosyl-(1→2)-O-[β-D-glucopyranosyl-(1→3)]-β-D-glucopyranoside, Jujubogenin 3-O-[α-L-arabinofuranosyl-(1→2)-O-[β-D-glucopyranosyl-(1→3)]-β-D-glucopyranoside]
About This Item
Productos recomendados
grade
analytical standard
Quality Level
assay
≥95% (HPLC)
shelf life
limited shelf life, expiry date on the label
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
application(s)
food and beverages
format
neat
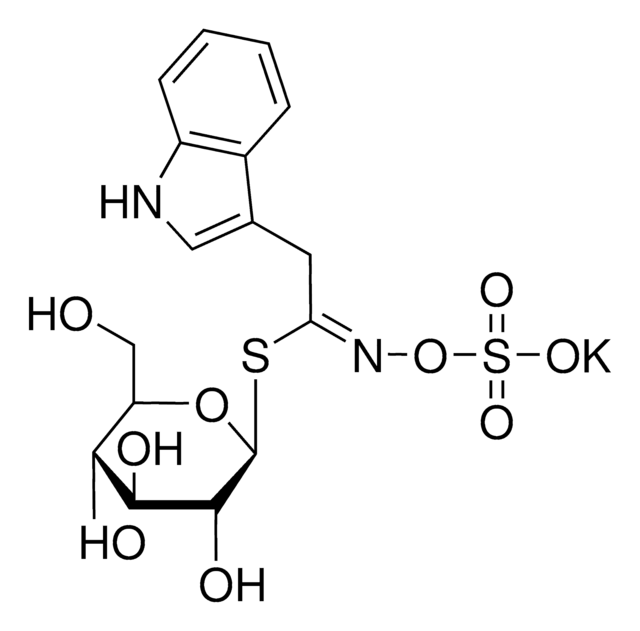
SMILES string
C\C(C)=C\[C@H]1C[C@](C)(O)[C@@H]2[C@H]3CC[C@@H]4[C@@]5(C)CC[C@H](O[C@@H]6O[C@H](CO)[C@@H](O)[C@H](O[C@@H]7O[C@H](CO)[C@@H](O)[C@H](O)[C@H]7O)[C@H]6O[C@@H]8O[C@@H](CO)[C@H](O)[C@H]8O)C(C)(C)[C@@H]5CC[C@@]4(C)[C@@]39CO[C@@]2(C9)O1
InChI
1S/C47H76O18/c1-21(2)14-22-15-45(7,57)38-23-8-9-28-43(5)12-11-29(42(3,4)27(43)10-13-44(28,6)46(23)19-47(38,65-22)58-20-46)62-41-37(64-39-34(55)31(52)25(17-49)60-39)36(32(53)26(18-50)61-41)63-40-35(56)33(54)30(51)24(16-48)59-40/h14,22-41,48-57H,8-13,15-20H2,1-7H3/t22-,23+,24+,25-,26+,27-,28+,29-,30+,31-,32+,33-,34+,35+,36-,37+,38-,39-,40-,41-,43-,44+,45-,46-,47-/m0/s1
InChI key
CDEVGTJBRPBOPH-INTDMYAHSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
Packaging
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico