B3253
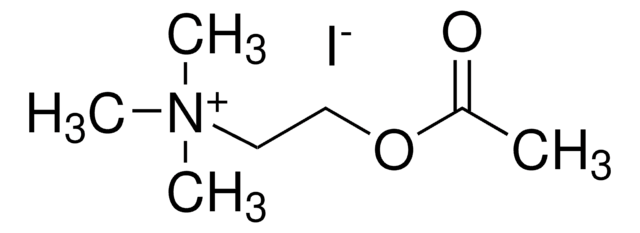
Butyrylthiocholine iodide
≥98%
Sinónimos:
(2-Mercaptoethyl)trimethylammonium iodide butyrate
About This Item
Productos recomendados
Quality Level
assay
≥98%
form
powder
mp
171-174 °C (lit.)
storage temp.
−20°C
SMILES string
[I-].CCCC(=O)SCC[N+](C)(C)C
InChI
1S/C9H20NOS.HI/c1-5-6-9(11)12-8-7-10(2,3)4;/h5-8H2,1-4H3;1H/q+1;/p-1
InChI key
WEQAAFZDJROSBF-UHFFFAOYSA-M
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
Application
- Label-Free and Ultrasensitive Detection of Butyrylcholinesterase: A study demonstrated the use of Mn(II)-based electron spin resonance spectroscopy for the ultrasensitive detection of butyrylcholinesterase, using Butyrylthiocholine iodide as a substrate to quantify enzyme activity in the presence of organophosphorus pesticides, crucial for biochemical assay applications (Tang et al., 2022).
- Novel Nanozyme for Biosensing: Research developed a Co, N co-doped porous carbon-based nanozyme, demonstrating its utility as an oxidase mimic for fluorescence and colorimetric biosensing of butyrylcholinesterase, employing Butyrylthiocholine iodide as a key substrate, relevant in enzyme kinetics analysis (Sun et al., 2022).
- Detection System for Anti-Alzheimer′s Drug Screening: A fluorescent platform was constructed using copper nanoclusters and MnO2 nanosheets for the detection of butyrylcholinesterase activity, utilizing Butyrylthiocholine iodide, which may facilitate the screening of anti-Alzheimer′s drugs and probe cholinergic system interactions (Chen et al., 2022).
- Dual-Channel Detection of Butyrylcholinesterase: A study introduced bifunctional metal-organic frameworks with integrated fluorescence and oxidase activities, developed for dual-channel detection of butyrylcholinesterase using Butyrylthiocholine iodide, enhancing methodologies in biochemical assays (Wang et al., 2022).
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico