275581
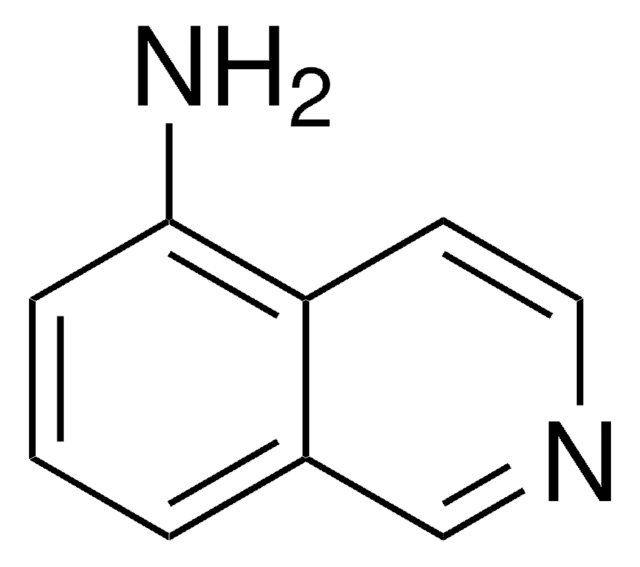
6-Aminoquinoline
98%
Sinónimos:
6-Quinolinamine
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C9H8N2
Número de CAS:
Peso molecular:
144.17
Beilstein:
113320
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Ensayo
98%
Formulario
solid
bp
146 °C/0.3 mmHg (lit.)
mp
115-119 °C (lit.)
cadena SMILES
Nc1ccc2ncccc2c1
InChI
1S/C9H8N2/c10-8-3-4-9-7(6-8)2-1-5-11-9/h1-6H,10H2
Clave InChI
RJSRSRITMWVIQT-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Aplicación
6-Aminoquinoline was used as an internal standard in determining serum nicotine and cotinine simultaneously by using high-performance liquid chromatography (HPLC)-fluorometric detection with a postcolumn ultraviolet-photoirradiation system. It was also used as a fluorescent derivatizing agent for the detection of biochemicals and in the synthesis of tertiary N-methylated enaminonesa.
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Jong Bae Seo et al.
PloS one, 10(12), e0144432-e0144432 (2015-12-15)
Hydrolysis of phosphatidylinositol 4,5-bisphosphate (PIP2) of the plasma membrane by phospholipase C (PLC) generates two critical second messengers, inositol-1,4,5-trisphosphate and diacylglycerol. For the enzymatic reaction, PIP2 binds to positively charged amino acids in the pleckstrin homology domain of PLC. Here
S A Cohen et al.
Analytical biochemistry, 211(2), 279-287 (1993-06-01)
A highly reactive amine derivatizing reagent, 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate, has been synthesized. In a rapid, one-step procedure, the compound reacts with amino acids to form stable unsymmetric urea derivatives which are readily amenable to analysis by reversed phase HPLC. Studies on
W Nashabeh et al.
Journal of chromatography, 600(2), 279-287 (1992-05-29)
The electrophoretic behavior of derivatized linear and branched oligosaccharides from various sources was examined in capillary zone electrophoresis with polyether-coated fused-silica capillaries. Two UV-absorbing (also fluorescent) derivatizing agents (2-aminopyridine and 6-aminoquinoline) were utilized for the electrophoresis and sensitive detection of
Bryan P Ruddy et al.
Journal of controlled release : official journal of the Controlled Release Society, 306, 83-88 (2019-06-01)
Subcutaneous delivery of nicotine was performed using a novel electrically-operated needle-free jet injector, and compared to hypodermic needle delivery in a porcine model. Nicotine was delivered as a single, one-milligram dose into the abdominal skin, formulated as a 50 microliter
Pierre-Jean Aragon et al.
Chemical & pharmaceutical bulletin, 52(6), 659-663 (2004-06-10)
Indoloquinoline alkaloid cryptolepine and pyridocarbazole alkaloid ellipticine are of great interest because in vitro and in vivo studies revealed their good cytotoxic properties. In order to obtain some biologically active analogs of these compounds, we developped a synthesis based on
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico