M9929
Methotrexate
meets USP testing specifications
Synonym(s):
Methotrexate hydrate, 4-Amino-10-methylfolic acid hydrate, L-4-Amino-N10-methylpteroylglutamic acid hydrate, L-Amethopterin hydrate, Antifolan hydrate, MTX hydrate, Methylaminopterin hydrate
About This Item
Recommended Products
biological source
synthetic
Quality Level
Agency
USP/NF
meets USP testing specifications
Assay
98.0-102.0%
form
powder
storage condition
(Tightly closed. Dry. Keep in a well-ventilated place. )
technique(s)
activity assay: suitable
cell based assay: suitable
impurities
≤0.1% Residue on ignition (Ash)
≤12.0% water (Karl Fischer)
color
yellow to orange-yellow
mp
185-204 °C (365 - 399 °F)
solubility
water: insoluble
antibiotic activity spectrum
parasites
application(s)
advanced drug delivery
metabolomics
microbiology
Mode of action
DNA synthesis
storage temp.
−20°C
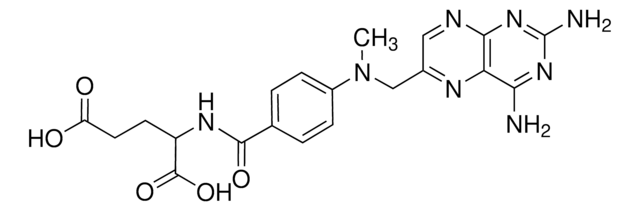
SMILES string
[H]O[H].CN(Cc1cnc2nc(N)nc(N)c2n1)c3ccc(cc3)C(=O)N[C@@H](CCC(O)=O)C(O)=O
InChI
1S/C20H22N8O5.H2O/c1-28(9-11-8-23-17-15(24-11)16(21)26-20(22)27-17)12-4-2-10(3-5-12)18(31)25-13(19(32)33)6-7-14(29)30;/h2-5,8,13H,6-7,9H2,1H3,(H,25,31)(H,29,30)(H,32,33)(H4,21,22,23,26,27);1H2/t13-;/m0./s1
InChI key
FPJYMUQSRFJSEW-ZOWNYOTGSA-N
Gene Information
human ... DHFR(1719) , DHFRP1(573971) , FPGS(2356) , SLC19A1(6573) , TYMS(7298)
mouse ... Dhfr(13361)
rat ... Dhfr(24312)
Looking for similar products? Visit Product Comparison Guide
Application
Biochem/physiol Actions
Other Notes
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Oral - Eye Irrit. 2 - Repr. 1B - Skin Irrit. 2
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Cancer research innovations address the complexity of the disease, providing advanced technologies for varied applications.
Cancer research innovations address the complexity of the disease, providing advanced technologies for varied applications.
Biofiles reviews innovative technologies for cancer research, reflecting the complexity of the disease.
Cancer research innovations address the complexity of the disease, providing advanced technologies for varied applications.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service