T89702
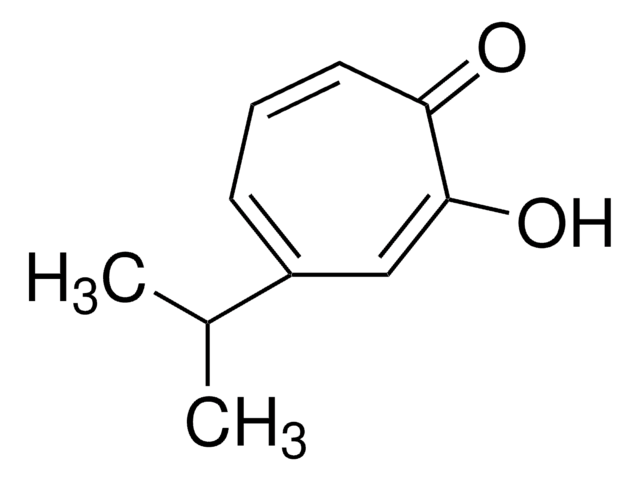
Tropolone
98%
Synonym(s):
2-Hydroxy-2,4,6-cycloheptatrien-1-one
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C7H6O2
CAS Number:
Molecular Weight:
122.12
Beilstein:
1904978
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
bp
80-84 °C/0.1 mmHg (lit.)
mp
50-52 °C (lit.)
storage temp.
2-8°C
SMILES string
OC1=CC=CC=CC1=O
InChI
1S/C7H6O2/c8-6-4-2-1-3-5-7(6)9/h1-5H,(H,8,9)
InChI key
MDYOLVRUBBJPFM-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Tropolone is a sensitive reagent for reducing sugars. It is also an organic chelator that can form complexes with trivalent lathanide ions (Eu3+, Gd3+, and Tb3+).
It can also be used as:
It can also be used as:
- A precursor to synthesize azulene derivatives such as methyl 2-methylazulene-1-carboxylate.
- A reagent to prepare fused heterocycles and complexes of Ga(III) and In(III).
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Skin Corr. 1B - Skin Sens. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
233.6 °F - closed cup
Flash Point(C)
112 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Spectroscopy and non-radiative processes in Gd3+, Eu3+ and Tb3+ tropolonates.
Santos BS, et al.
Spectrochimica Acta. Part A, Molecular and Biomolecular Spectroscopy, 54(13), 2237-2245 (1998)
Trends Heterocycl. Chem., 1, 137-137 (1990)
Structural characterization of two lipophilic tris(tropolonato) gallium(III) and indium(III) complexes of radiopharmaceutical interest
F.Nepveu and F.JasanadaL.Walz
Inorgorganica Chimica Acta, 211, 141-141 (1993)
Eric Sabondjian et al.
Contrast media & molecular imaging, 7(1), 76-84 (2012-02-22)
A challenge with cardiac cell therapy is determining the location of cells relative to infarct tissue. As cells are viable following ¹¹¹In-labeling, and first-pass CT imaging can identify regions of myocardial infarction, we evaluated the feasibility of a SPECT/CT system
Jack Davison et al.
Proceedings of the National Academy of Sciences of the United States of America, 109(20), 7642-7647 (2012-04-18)
A gene cluster encoding the biosynthesis of the fungal tropolone stipitatic acid was discovered in Talaromyces stipitatus (Penicillium stipitatum) and investigated by targeted gene knockout. A minimum of three genes are required to form the tropolone nucleus: tropA encodes a
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service