P24055
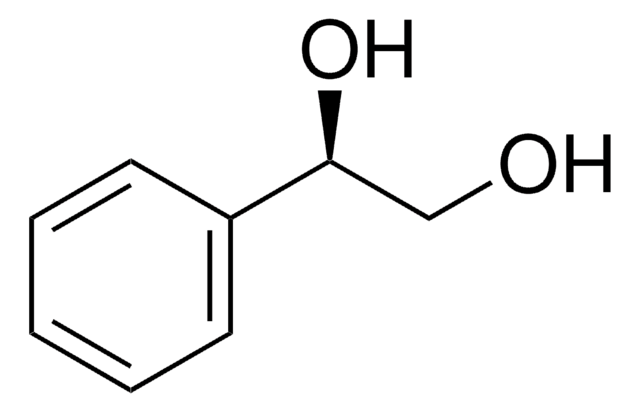
1-Phenyl-1,2-ethanediol
97%
Synonym(s):
(±)-1-Phenyl-1,2-ethanediol, (±)-Phenylethylene glycol
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
C6H5CH(OH)CH2OH
CAS Number:
Molecular Weight:
138.16
Beilstein:
1306723
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
bp
272-274 °C/760 mmHg (lit.)
mp
66-68 °C (lit.)
SMILES string
OCC(O)c1ccccc1
InChI
1S/C8H10O2/c9-6-8(10)7-4-2-1-3-5-7/h1-5,8-10H,6H2
InChI key
PWMWNFMRSKOCEY-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Ashwini C Mathpati et al.
Journal of biotechnology, 262, 1-10 (2017-09-30)
Kinetic resolution of rac-1,2-diols using the biocatalyst Burkholderia cepacia lipase (BCL) immobilized on a biodegradable binary blend support of hydroxypropyl methyl cellulose(HPMC)/polyvinyl alcohol (PVA) has been investigated. The immobilization technique improved enzyme activity significantly and it has excellent recyclability with
Yao Nie et al.
Organic & biomolecular chemistry, 9(11), 4070-4078 (2011-04-21)
The application of biocatalysis to the synthesis of chiral molecules is one of the greenest technologies for the replacement of chemical routes due to its environmentally benign reaction conditions and unparalleled chemo-, regio- and stereoselectivities. We have been interested in
Lingyun Rui et al.
Applied and environmental microbiology, 71(7), 3995-4003 (2005-07-08)
DNA shuffling and saturation mutagenesis of positions F108, L190, I219, D235, and C248 were used to generate variants of the epoxide hydrolase of Agrobacterium radiobacter AD1 (EchA) with enhanced enantioselectivity and activity for styrene oxide and enhanced activity for 1,2-epoxyhexane
Yanbin Liu et al.
Journal of industrial microbiology & biotechnology, 33(4), 274-282 (2005-12-02)
A microorganism with the ability to catalyze the resolution of racemic phenyloxirane was isolated and identified as Aspergillus niger SQ-6. Chiral capillary electrophoresis was successfully applied to separate both phenyloxirane and phenylethanediol. The epoxide hydrolase (EH) involved in this resolution
Li Cao et al.
Biotechnology and bioengineering, 94(3), 522-529 (2006-02-25)
Soluble epoxide hydrolase (EH) from the potato Solanum tuberosum and an evolved EH of the bacterium Agrobacterium radiobacter AD1, EchA-I219F, were purified for the enantioconvergent hydrolysis of racemic styrene oxide into the single product (R)-1-phenyl-1,2-ethanediol, which is an important intermediate
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service