522910
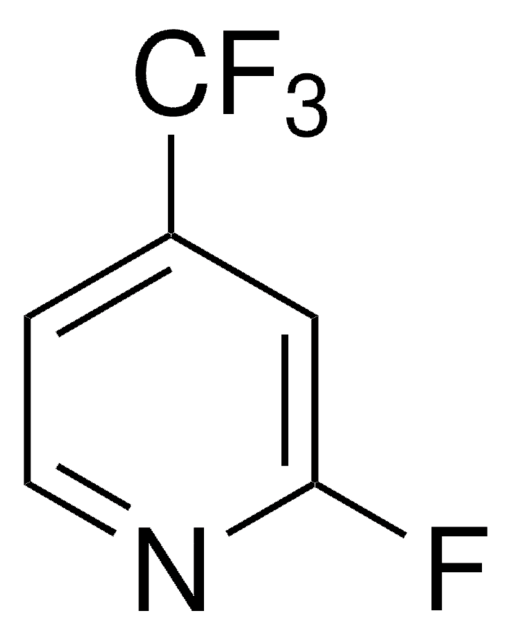
4-(Trifluoromethyl)pyridine
97%
Synonym(s):
4-(Trifluoromethyl)pyridine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C6H4F3N
CAS Number:
Molecular Weight:
147.10
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
refractive index
n20/D 1.417 (lit.)
bp
110 °C (lit.)
density
1.27 g/mL at 25 °C (lit.)
functional group
fluoro
SMILES string
FC(F)(F)c1ccncc1
InChI
1S/C6H4F3N/c7-6(8,9)5-1-3-10-4-2-5/h1-4H
InChI key
IIYVNMXPYWIJBL-UHFFFAOYSA-N
General description
4-(Trifluoromethyl)pyridine is a pyridine derivative. It can be prepared by trifluoromethylation of 4-iodobenzene.
Application
4-(Trifluoromethyl)pyridine may be used in the following:
- Preparation of (trifluoromethyl)pyridyllithiums, via metalation reaction.
- Synthesis of metal-organic frameworks (MOFs).
- Synthesis of methiodide salts.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Flam. Liq. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
68.0 °F - closed cup
Flash Point(C)
20 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Manfred Schlosser et al.
Chemical Society reviews, 36(7), 1161-1172 (2007-06-20)
Pyridines carrying heterosubstituents (such as carboxy, amido, amino, alkoxy or trifluoromethyl groups or solely individual halogen atoms) can be readily and site selectively metalated. Subsequent reaction with a suitable electrophile opens rational access to a wealth of new building blocks
Fluorinated pyridine derivatives: Part 1. The synthesis of some mono-and bis-quaternary pyridine salts of potential use in the treatment of nerve agent poisoning.
Timperley CM, et al.
Journal of Fluorine Chemistry, 126(8), 1160-1165 (2005)
Enhancement of CO2/N2 selectivity in a metal-organic framework by cavity modification.
Bae YS, et al.
Journal of Materials Chemistry, 19(15), 2131-2134 (2009)
Joonhyuck Park et al.
ACS nano, 14(9), 11579-11593 (2020-08-14)
A heat-up method for quantum dots (QDs) synthesis holds distinctive benefits for large-scale production with its simplicity, scalability, and high reproducibility. Its applications, however, have been limited because it inevitably yields a strong overlap between the nucleation and the growth
Copper-mediated trifluoromethylation of heteroaromatic compounds by trifluoromethyl sulfonium salts.
Cheng-Pan Zhang et al.
Angewandte Chemie (International ed. in English), 50(8), 1896-1900 (2011-02-18)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service