All Photos(1)
About This Item
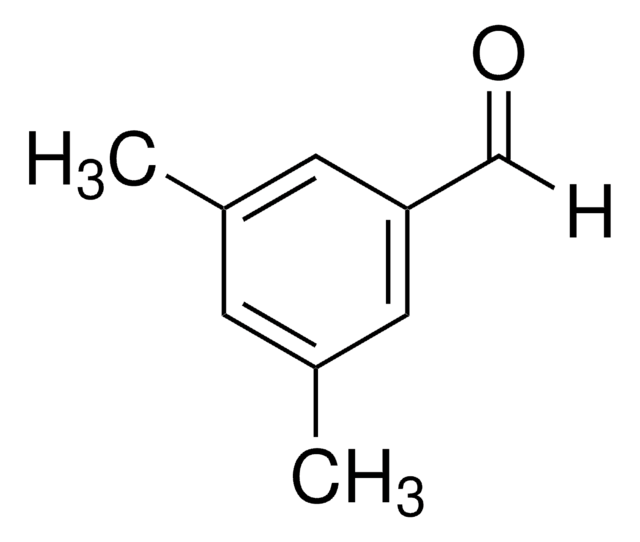
Linear Formula:
(CH3)2C6H3CHO
CAS Number:
Molecular Weight:
134.18
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
refractive index
n20/D 1.551 (lit.)
bp
226 °C (lit.)
density
1.012 g/mL at 25 °C (lit.)
functional group
aldehyde
SMILES string
Cc1ccc(C=O)cc1C
InChI
1S/C9H10O/c1-7-3-4-9(6-10)5-8(7)2/h3-6H,1-2H3
InChI key
POQJHLBMLVTHAU-UHFFFAOYSA-N
General description
3,4-Dimethylbenzaldehyde (3,4-DMB) is a benzaldehyde derivative. It is the OH radical initiated oxidative degradation product of trimethylbenzene. The rate coefficient of the gas-phase reaction between 3,4-DMB and OH radical is 24.6±4.0×10-12cm3molecule-1s-1. The vibrational analysis of 3,4-DMB based on FT-IR spectra, FT-Raman spectra, ab initio and density functional theory (DFT) calculations have been reported. It is formed as an intermediate during the transformation of furfural into gasoline-range fuels using ZSM(Zeolite Socony Mobil)-5-based catalysts.
Application
3,4-Dimethylbenzaldehyde may be used in the synthesis of 3,4-dimethylmethcathinone (DMMC) and 3,4-dimethyl-dibenzylidene sorbitol.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Synthesis of 3,4-Dimethyl-dibenzylidene Sorbitol at Room Temperature.
Yin ZY, et al.
Huaxue Shiji, 47(3), 174-174 (2006)
Catalytic fast pyrolysis of furfural over H-ZSM-5 and Zn/H-ZSM-5 catalysts.
Fanchiang WL and Lin YC.
Applied Catalysis A: General, 419, 102-110 (2012)
Urinary excretion and metabolism of the newly encountered designer drug 3,4-dimethylmethcathinone in humans.
Shima N, et al.
Forensic Toxicology, 31(1), 101-112 (2013)
Rate coefficients for the gas-phase reaction of hydroxyl radicals with the dimethylbenzaldehydes.
Clifford GM and Wenger JC.
International Journal of Chemical Kinetics, 38(9), 563-569 (2006)
Vibrational spectroscopy investigation using ab initio and density functional theory analysis on the structure of 3, 4-dimethylbenzaldehyde.
Sundaraganesan N, et al.
Spectrochimica Acta. Part A, Molecular and Biomolecular Spectroscopy, 68(3), 680-687 (2007)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service