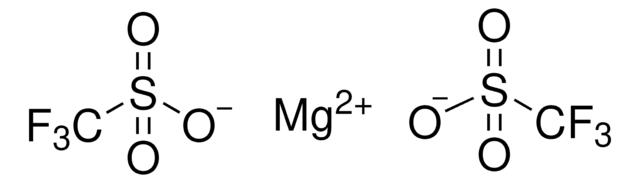422843
Potassium trifluoromethanesulfonate
98%
Synonym(s):
Potassium triflate, Trifluoromethanesulfonic acid potassium salt
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
CF3SO3K
CAS Number:
Molecular Weight:
188.17
Beilstein:
3727495
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
powder
functional group
fluoro
triflate
SMILES string
[K+].[O-]S(=O)(=O)C(F)(F)F
InChI
1S/CHF3O3S.K/c2-1(3,4)8(5,6)7;/h(H,5,6,7);/q;+1/p-1
InChI key
GLGXXYFYZWQGEL-UHFFFAOYSA-M
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Potassium trifluoromethanesulfonate (KOTf, potassium triflate) is the potassium salt of trifluoromethanesulphonic acid. It has been prepared by neutralizing a warm aqueous solution of trifluoromethanesulphonic acid with potassium carbonate. The structure of siloxane–poly(oxyethylene) hybrids doped with potassium triflate has been investigated.
Application
Potassium trifluoromethanesulfonate (potassium triflate) is suitable as a reagent in the synthesis of guanine-quadruplex hybrid materials by the self-assembly of a guanine-siloxane monomer.
It may be used in the following studies:
It may be used in the following studies:
- As a template source for constructing the heterotopic isothiosemicarbazide-based macrocyclic ligand.
- As a supporting electrolyte in the electrochemical study of evidence for gold anion in ethylenediamine.
- As a reagent in the synthesis of imidazolium salt, 3-methyl-1-(3R,3aR,6S,6aR)-[6-(benzyloxy)-hexahydrofuro[3,2-b]furan-3-yl]imidazolium trifluoromethanesulfonate.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Gregory D Bowden et al.
Scientific reports, 9(1), 11370-11370 (2019-08-08)
Recent advancements in 18F radiochemistry, such as the advent of copper-mediated radiofluorination (CMRF) chemistry, have provided unprecedented access to novel chemically diverse PET probes; however, these multicomponent reactions have come with a new set of complex optimization problems. Design of
Evidence for the gold anion in ethylenediamine.
Jagannathan R, et al.
Inorganic Chemistry, 24(1), 113-114 (1985)
Potassium-controlled synthesis of heterotopic macrocycles based on isothiosemicarbazide.
Arion VB, et al.
Inorgorganica Chimica Acta, 328(1), 123-133 (2002)
Properties and reactivities of pentadentate ethylenediaminetetraacetate complexes of ruthenium (III) and-(II).
Matsubara T and Creutz C.
Inorganic Chemistry, 18(7), 1956-1966 (1979)
Sara Santiago-Malagón et al.
Biosensors & bioelectronics, 175, 112879-112879 (2020-12-15)
One of the limitations of many skin-patch wearable sensors today is their dependence on silicon-based electronics, increasing their complexity and unit cost. Self-powered sensors, in combination with electrochromic materials, allow simplifying the construction of these devices, leading to powerful analytical
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service