D115207
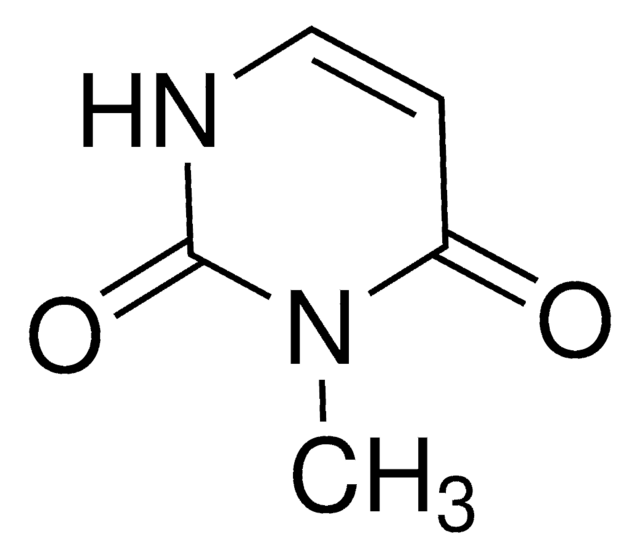
6-Methyluracil
97%
Synonym(s):
2,4-Dihydroxy-6-methylpyrimidine
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C5H6N2O2
CAS Number:
Molecular Weight:
126.11
Beilstein:
115647
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
powder
mp
318 °C (dec.) (lit.)
SMILES string
CC1=CC(=O)NC(=O)N1
InChI
1S/C5H6N2O2/c1-3-2-4(8)7-5(9)6-3/h2H,1H3,(H2,6,7,8,9)
InChI key
SHVCSCWHWMSGTE-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Repr. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Lucie Brulíková et al.
Carbohydrate research, 346(14), 2136-2144 (2011-08-26)
A number of 5-alkoxymethyluracil analogues were synthesized to evaluate their cytotoxic activity. 5-Alkoxymethyluracil derivatives 1 were prepared via known nucleophilic substitution of 5-chloromethyluracil 5 and subsequently transformed to their corresponding nucleosides 2. All prepared compounds were submitted to cytotoxic activity
Xiao Lu et al.
Bioorganic & medicinal chemistry, 15(23), 7399-7407 (2007-09-18)
Novel compounds 1a-u, which can be considered as hybrid analogues of MKC-442 and pyridinon, have been synthesized and evaluated as inhibitors of HIV-1 reverse transcriptase (HIV-1 RT). Starting from 6-methyluracil 2, 1-alkylated-5-bromomethyl-6-methyluracils 8 was prepared in four steps by hydroxymethylation
M Iu Gerasimenko et al.
Voprosy kurortologii, fizioterapii, i lechebnoi fizicheskoi kultury, (2)(2), 27-31 (2006-06-07)
Clinical, biochemical, experimental, heat physical studies and mathematical calculations have shown that combined effect of drug-laser can be differentiated into the following effects: photophoresis, light pressure and photodynamic effect.
Fabio C Tucci et al.
Chirality, 17(9), 559-564 (2005-10-01)
1-(2,6-Difluorobenzyl)-3-[(2R)-amino-2-phenethyl]-5-(2-fluoro-3-methoxyphenyl)-6-methyluracil (6), a potent and orally active antagonist of the human gonadotropin-releasing hormone receptor, exists as a pair of atropisomers in solution, which was detected by NMR spectroscopy, and separable by HPLC. In addition to a (R)-configured benzylamine, there is
[Surgical infected wound treatment in patients with severe gastrointestinal ulcer hemorrhage].
V V Ganzhiĭ
Klinichna khirurhiia, (11-12)(11-12), 18-19 (2003-01-29)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service



![2-{[1-(tert-Butoxycarbonyl)-4-piperidinyl]sulfonyl}benzoic acid AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/224/106/eee847a5-ad90-45e6-b100-f59ed97f8957/640/eee847a5-ad90-45e6-b100-f59ed97f8957.png)





