453757
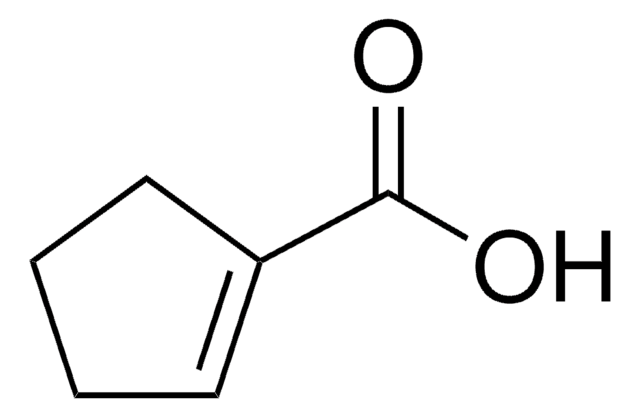
3-Cyclohexene-1-carboxylic acid
97%
Synonym(s):
1,2,3,6-Tetrahydrobenzoic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
C6H9CO2H
CAS Number:
Molecular Weight:
126.15
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
refractive index
n20/D 1.48 (lit.)
bp
130-133 °C/4 mmHg (lit.)
mp
17 °C (lit.)
density
1.081 g/mL at 25 °C (lit.)
functional group
carboxylic acid
SMILES string
OC(=O)C1CCC=CC1
InChI
1S/C7H10O2/c8-7(9)6-4-2-1-3-5-6/h1-2,6H,3-5H2,(H,8,9)
InChI key
VUSWCWPCANWBFG-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
230.0 °F - closed cup
Flash Point(C)
110 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Yi Yang et al.
Environmental science & technology, 48(4), 2344-2351 (2014-02-01)
The effect of halides on organic contaminant destruction efficiency was compared for UV/H2O2 and UV/S2O8(2-) AOP treatments of saline waters; benzoic acid, 3-cyclohexene-1-carboxylic acid, and cyclohexanecarboxylic acid were used as models for aromatic, alkene, and alkane constituents of naphthenic acids
Bromination of 3-cyclohexene-1-carboxylic acid, epoxydation of methyl 3-cyclohexene-1-carboxylate and opening of methyl cis-and trans-3, 4-epoxycyclohexane-1-carboxylate: Stereochemical results.
Bellucci G, et al.
Tetrahedron, 28(13), 3393-3399 (1972)
The metabolism of cyclohexanecarboxylic acid and 3-cyclohexenecarboxylic acid by Pseudomonas putida.
E R Blakley et al.
Canadian journal of microbiology, 28(12), 1324-1329 (1982-12-01)
A strain of Pseudomonas putida grew rapidly on cyclohexanecarboxylic acid as a sole source of carbon. A CoA-mediated beta-oxidation pathway was induced for the metabolism of the compound. The organism could not utilize 3-cyclohexenecarboxylic acid as a sole source of
Karine Barral et al.
Journal of medicinal chemistry, 48(2), 450-456 (2005-01-22)
Starting from commercially available (rac)-3-cyclohexene-1-carboxylic acid, a series of purine and pyrimidine cis-substituted cyclohexenyl and cyclohexanyl nucleosides were synthesized through a key Mitsunobu reaction. Antiviral evaluations were performed on HIV, coxsackie B3, and herpes viruses (HSV-1, HSV-2, VZV, HCMV). Three
Pia Haslund Jørgensen et al.
Contact dermatitis, 56(3), 146-150 (2007-02-14)
Hydroxyisohexyl-3-cyclohexene carboxaldehyde (HICC) known as Lyral is a frequent allergen. It is used in more than 50% of marketed deodorants. The aim of the present study was to determine elicitation thresholds for HICC under simulated conditions of deodorant use. 15
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service