All Photos(1)
About This Item
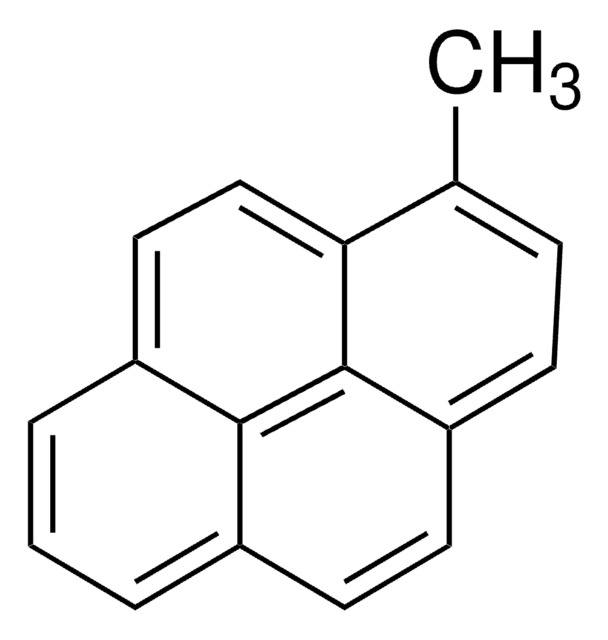
Empirical Formula (Hill Notation):
C18H12O
CAS Number:
Molecular Weight:
244.29
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
solid
mp
86-89 °C (lit.)
SMILES string
CC(=O)c1ccc2ccc3cccc4ccc1c2c34
InChI
1S/C18H12O/c1-11(19)15-9-7-14-6-5-12-3-2-4-13-8-10-16(15)18(14)17(12)13/h2-10H,1H3
InChI key
KCIJNJVCFPSUBQ-UHFFFAOYSA-N
General description
1-Acetylpyrene is a pyrene derivative. Its synthesis has been reported. Its phytophysical properties have been studied using absolute fluorescence quantum yield measurement and time-dependent density functional theory (TD-DFT) calculations. Its ability to interact with human cytochromes P450 2A13, 2A6, and 1B1 and enzyme inhibition has been reported. Its function as an environment-sensitive fluorophore has been investigated.
Application
1-Acetylpyrene is suitable for use in a comparative study on the photoinitiating efficiency of pyrene, 1-acetylpyrene and 1-(bromoacetyl)pyrene for copolymerization of styrene with acrylonitrile. It may be used in the following studies:
- As a starting material in the synthesis of ethynlypyrene. 1-(1-chlorovinyl)pyrene was also isolated during this reaction.
- As a starting material in the synthesis of substituted pyrene derivatives incorporated heterocyclic and sugar moieties.
- Synthesis of (E)-pyrene oxime ester conjugates of carboxylic acids.
- Synthesis of tertiary alcohols based on 1-acetylpyrene.
- Synthesis of (E)-N-[1-(pyren-1-yl)ethylidene]chrysene-2-amine.
- Synthesis of 3,3-di(methylsulfanyl)-1-(1-pyrenyl)-2-propen-1-one.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
H Surya Prakash Rao et al.
Beilstein journal of organic chemistry, 3, 31-31 (2007-10-02)
The cycloaddition of the von Leusen's reagent (p-tolylsulfonyl)methyl isocyanide (TosMIC) to alpha-aroylketene dithioacetals (AKDTAs) in the presence of sodium hydride in THF at rt resulted in a facile synthesis of the 4-aroyl-3-methylsulfanyl-2-tosylpyrroles 3 in good yield along with a minor
Tsutomu Shimada et al.
Chemical research in toxicology, 26(4), 517-528 (2013-02-26)
A total of 68 chemicals including derivatives of naphthalene, phenanthrene, fluoranthene, pyrene, biphenyl, and flavone were examined for their abilities to interact with human P450s 2A13 and 2A6. Fifty-one of these 68 chemicals induced stronger Type I binding spectra (iron
One-pot synthesis of 1-acetylpyrene over supported phosphotungstic heteropoly acid catalysts.
He MQ, et al.
Reaction Kinetics, Mechanisms and Catalysis, 108(2), 531-544 (2013)
Synthesis, characterization and pharmacological investigations of some novel heterocyclic derivatives incorporating pyrene and sugar moieties.
Khalifa NM, et al.
Research on Chemical Intermediates, 40(4), 1565-1574 (2014)
Tertiary alcohols based on 1-acetylpyrene.
Dikusar EA.
Russ. J. Org. Chem., 43(10), 1492-1494 (2007)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service