All Photos(1)
About This Item
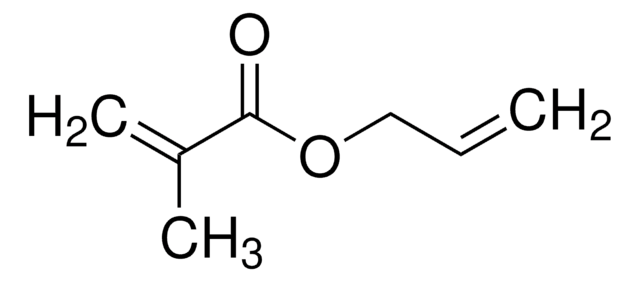
Linear Formula:
C6H5CH=CHCOOCH3
CAS Number:
Molecular Weight:
162.19
Beilstein:
386468
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥99.0% (GC)
form
crystals
bp
260-262 °C (lit.)
mp
33-38 °C (lit.)
functional group
ester
phenyl
SMILES string
COC(=O)\C=C\c1ccccc1
InChI
1S/C10H10O2/c1-12-10(11)8-7-9-5-3-2-4-6-9/h2-8H,1H3/b8-7+
InChI key
CCRCUPLGCSFEDV-BQYQJAHWSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Kathrin Fink et al.
Journal of agricultural and food chemistry, 52(10), 3065-3068 (2004-05-13)
For the authenticity assessment of (E)-methyl cinnamate from different origins, combustion/pyrolysis-isotope ratio mass spectrometry (C/P-IRMS) was used by an elemental analyzer (EA) and on-line capillary gas chromatography coupling (HRGC-C/P-IRMS). For that reason, (E)-methyl cinnamate self-prepared from synthetic, natural, and semisynthetic
Bhanu Prakash et al.
International journal of food microbiology, 153(1-2), 183-191 (2011-12-06)
The investigation deals with antifungal, antiaflatoxin and antioxidant efficacy of Zanthoxylum alatum Roxb. essential oil (EO), its two major constituents and their comparison with five commonly used organic acid preservatives. The chemical profile of EO, characterized through GC and GC-MS
L P Delbressin et al.
Archives of toxicology, 49(3-4), 321-330 (1982-03-01)
The urinary mercapturic acid excretion by female rats of methyl atropate (alpha-phenyl methyl acrylate) and methyl cinnamate (beta-phenyl methyl acrylate) has been studied. On the basis of the structures of these mercapturic acids the conclusion can be drawn that these
K Shimoi et al.
Mutation research, 146(1), 15-22 (1985-07-01)
UV-induced mutagenesis in Escherichia coli B/r WP2 was enhanced by certain derivatives of methyl cinnamate which themselves were not mutagenic. Methyl ferulate, methyl isoferulate and methyl sinapate showed this effect markedly. Such an enhancement effect was absent with the derivatives
Takuo Sawahata et al.
Mycorrhiza, 18(2), 111-114 (2007-12-11)
Two major volatiles produced by the mycelia and fruiting bodies of Tricholoma matsutake (1-octen-3-ol and methyl cinnamate) repel a mycophagous collembolan, Proisotoma minuta. Aggregation of the collembolans on their diet was significantly inhibited by exposure to 1 ppm methyl cinnamate
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service