15256
BSA + TMCS
for GC derivatization, LiChropur™, 93.0-97.0% (GC)
Synonyme(s) :
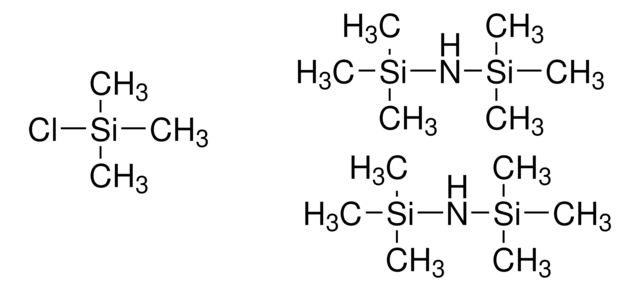
Bis(trimethylsilyl)acetamide + Trimethylchlorosilane
About This Item
Produits recommandés
Qualité
for GC derivatization
LiChropur™
Niveau de qualité
Pureté
93.0-97.0% (GC)
Composition
trimethylchlorosilane (minor component), 3.0-5.0% GC
Pertinence de la réaction
reagent type: derivatization reagent
reaction type: Silylations
Technique(s)
gas chromatography (GC): suitable
Description générale
Application
Caractéristiques et avantages
- BSA+TMCS has good solvent properties and can function as a silylation reagent without additional solvents.
- Alternatively, the mixture is very soluble in most commonly used silylation solvents.
- This combination is extremely sensitive to moisture and should be handled under dry conditions.
Autres remarques
Informations légales
Produit(s) apparenté(s)
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 4 Oral - Flam. Liq. 2 - Skin Corr. 1A
Risques supp
Code de la classe de stockage
3 - Flammable liquids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
53.6 °F - closed cup
Point d'éclair (°C)
12 °C - closed cup
Équipement de protection individuelle
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Articles
Results of a study involving the ability few Fluka silylating reagents to form GC-MS-compatible trimethylsilylmethyl derivatives of NSAIDs
Results of a study involving the ability few Fluka silylating reagents to form GC-MS-compatible trimethylsilylmethyl derivatives of NSAIDs
Results of a study involving the ability few Fluka silylating reagents to form GC-MS-compatible trimethylsilylmethyl derivatives of NSAIDs
Results of a study involving the ability few Fluka silylating reagents to form GC-MS-compatible trimethylsilylmethyl derivatives of NSAIDs
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique