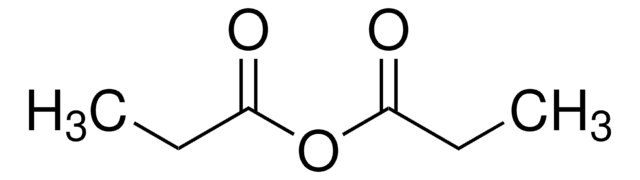
901233
(2R)-2-Phenyl-3,4-dihydro-2H-pyrimido[2,1-b][1,3]benzothiazole
≥95%
Synonyme(s) :
(R)-2-Phenyl-3,4-dihydro-2H-benzo[4,5]thiazolo[3,2-a]pyrimidine, (R)-Homobenzotetramisole, Birman (R)-HBTM
About This Item
Produits recommandés
Niveau de qualité
Pureté
≥95%
Forme
powder or chunks
Pf
145-147 °C
Groupe fonctionnel
phenyl
thioether
Température de stockage
2-8°C
Chaîne SMILES
C12=CC=CC=C1SC3=N[C@@H](C4=CC=CC=C4)CCN23
InChI
1S/C16H14N2S/c1-2-6-12(7-3-1)13-10-11-18-14-8-4-5-9-15(14)19-16(18)17-13/h1-9,13H,10-11H2/t13-/m1/s1
Clé InChI
ZMYZJAQMQBHNLH-CYBMUJFWSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Application
Autres remarques
Technology Spotlight: Homobenzotetramisole (HBTM): A General Organocatalyst for Asymmetric Acylations
[1] Determination of the Absolute Configuration β-Chiral Primary Alcohols Using the Competing Enantioselective Conversion Method
[2] Asymmetric Catalytic Synthesis of Thiochromenes via an Acyl Transfer-Initiated Cascade
[3] Rapid assembly of complex cyclopentanes employing chiral, α, β-unsaturated acylammonium intermediates
[4] Determination of Absolute Configuration Using Kinetic Resolution Catalysts
[5] Kinetic Resolution of Secondary Alcohols Using Amidine-Based Catalysts
Produit(s) apparenté(s)
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Certificats d'analyse (COA)
Vous ne trouvez pas la bonne version ?
Si vous avez besoin d'une version particulière, vous pouvez rechercher un certificat spécifique par le numéro de lot.
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Articles
We are proud to offer the isothiourea organocatalyst homobenzotetramisole (HBTM) as part of our asymmetric catalysis portfolio in both (R) and (S) enantiomeric forms.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique
![(2S)-2-Phenyl-2,3-dihydroimidazo[2,1-b][1,3]benzothiazole](/deepweb/assets/sigmaaldrich/product/structures/273/874/ec914181-6100-4322-aeb1-7665aaf7518c/640/ec914181-6100-4322-aeb1-7665aaf7518c.png)
![(2S)-2-Phenyl-3,4-dihydro-2H-pyrimido[2,1-b][1,3]benzothiazole 95%](/deepweb/assets/sigmaaldrich/product/structures/403/288/5e981722-19a3-4ad1-b2af-26137b4f5c18/640/5e981722-19a3-4ad1-b2af-26137b4f5c18.png)