374822
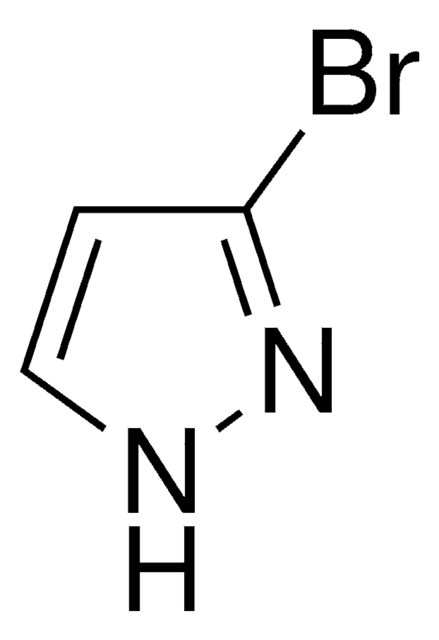
4-Bromopyrazole
99%
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Formule empirique (notation de Hill):
C3H3BrN2
Numéro CAS:
Poids moléculaire :
146.97
Numéro MDL:
Code UNSPSC :
12352100
ID de substance PubChem :
Nomenclature NACRES :
NA.22
Produits recommandés
Niveau de qualité
Pureté
99%
Forme
solid
Point d'ébullition
250-260 °C (lit.)
Pf
93-96 °C (lit.)
Groupe fonctionnel
bromo
Chaîne SMILES
Brc1cn[nH]c1
InChI
1S/C3H3BrN2/c4-3-1-5-6-2-3/h1-2H,(H,5,6)
Clé InChI
WVGCPEDBFHEHEZ-UHFFFAOYSA-N
Description générale
4-Bromopyrazole is a heteroaryl halide and its cyanation in the presence of palladium catalysts has been reported.
4-Bromopyrazole is a pyrazole derivative. It is reported to react with titanium tetrachloride to afford binary adducts. Mutagenicity of 4-bromopyrazole has been tested using the L-arabinose forward mutation assay of Salmonella typhimurium. It is reported to inhibit the oxidative phosphorylation, the ATP-32P exchange reaction, and energy dependent and independent calcium uptake.
Application
4-Bromopyrazole may be used in the preparation of 4-bromo-1-(2-chloroethyl)-1H-pyrazole. It may be used as starting material in the synthesis of 1,4′-bipyrazoles.
4-Bromopyrazole may be used in the preparation of solid hexacoordinate complexes by reaction with dimethyl- and divinyl-tindichloride.
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
dust mask type N95 (US), Eyeshields, Gloves
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Ilia A. Guzei et al.
Inorganic chemistry, 36(20), 4415-4420 (2001-10-24)
Treatment of titanium tetrachloride with 3,5-di-tert-butylpyrazole affords the complexes [3,5-(C(CH(3))(3))(2)C(3)H(3)N(2)](2)[TiCl(6)] and (3,5-(C(CH(3))(3))(2)C(3)HN(2))(2)TiCl(2) in 37 and 42% yields, respectively. An analogous reaction with 3,5-dimethylpyrazole, 3-methylpyrazole, 4-bromopyrazole, and 4-iodopyrazole leads to the formation of corresponding TiCl(4)L(2) binary adducts in 30-86% yields. Crystal
Kamal Nayan Sharma et al.
Dalton transactions (Cambridge, England : 2003), 42(11), 3908-3918 (2013-01-19)
The reactions of 4-bromo-1-(2-chloroethyl)-1H-pyrazole prepared from 4-bromopyrazole with the in situ generated PhSNa, PhSeNa, Na(2)S and Na(2)Se have resulted in thio/selenoether ligands L1-L4 respectively. The complexes [PdL1/L2Cl(2)](1-2) and [PdL3/L4Cl]BF(4) (3-4) of these ligands have been synthesized by reacting them with
Todd D Senecal et al.
Angewandte Chemie (International ed. in English), 52(38), 10035-10039 (2013-08-13)
Playing it safe: The nontoxic cyanide source K4 [Fe(CN)6]·3H2O can be used for the cyanation of (hetero)aryl halides. The application of palladacycle catalysts prevents poisoning during catalyst formation, thereby allowing for low catalyst loadings, fast reaction times, and wide heterocyclic
Effects of pyrazole, 4-bromopyrazole and 4-methylpyrazole on mitochondrial function.
A I Cederbaum et al.
Biochemical pharmacology, 23(2), 203-213 (1974-01-15)
Dichlorodialkyltin complexes with 4-bromopyrazole. The crystal structure of bis(4-bromopyrazole- N2)dichlorodimethyltin(IV).
Casellato U, et al.
Journal of Organometallic Chemistry, 486(1-2), 105-107 (1995)
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique