S5006
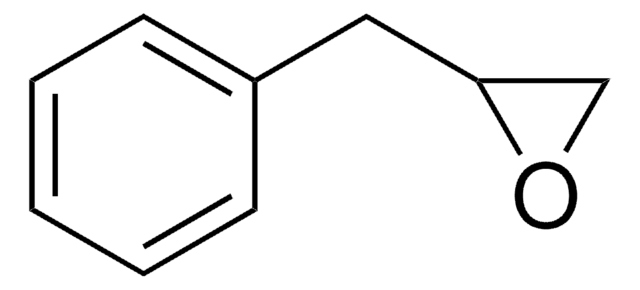
Styrene oxide
97%
Synonym(s):
1,2-Epoxyethylbenzene, Phenylethylene oxide, Phenyloxirane
About This Item
Recommended Products
vapor density
4.14 (vs air)
vapor pressure
<1 mmHg ( 20 °C)
Assay
97%
autoignition temp.
928 °F
expl. lim.
~22 %
refractive index
n20/D 1.535 (lit.)
bp
194 °C (lit.)
mp
−37 °C (lit.)
density
1.054 g/mL at 25 °C (lit.)
SMILES string
C1OC1c2ccccc2
InChI
1S/C8H8O/c1-2-4-7(5-3-1)8-6-9-8/h1-5,8H,6H2
InChI key
AWMVMTVKBNGEAK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Synthesis of poly (styrene oxide) with different molecular weights using tin catalysts: This study explores the ring-opening polymerization of styrene oxide using tin catalysts to produce homopolymers with varying molecular weights (Kayan, 2015).
- Electrogenerated BF3 from tetrafluoroborate-based ionic liquids: theoretical and experimental studies towards selective styrene oxide isomerization: Research on using electrogenerated BF3 to selectively isomerize styrene oxide, highlighting theoretical and experimental insights (Bortolami et al., 2021).
- Selective conversion of styrene oxide to 2-phenylethanol in cascade reactions over non-noble metal catalysts: This paper investigates the catalytic conversion of styrene oxide to 2-phenylethanol using non-noble metal catalysts (Sasu et al., 2016).
- Laboratory blueprints for interstellar searches of aromatic chiral molecules: rotational signatures of styrene oxide: Study of the rotational spectra of styrene oxide for potential detection in interstellar space (Stahl et al., 2020).
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Inhalation - Acute Tox. 4 Dermal - Carc. 1B - Eye Irrit. 2 - Muta. 1B - Skin Irrit. 2 - Skin Sens. 1
Storage Class Code
6.1C - Combustible, acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
176.0 °F - closed cup
Flash Point(C)
80 °C - closed cup
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service