670871
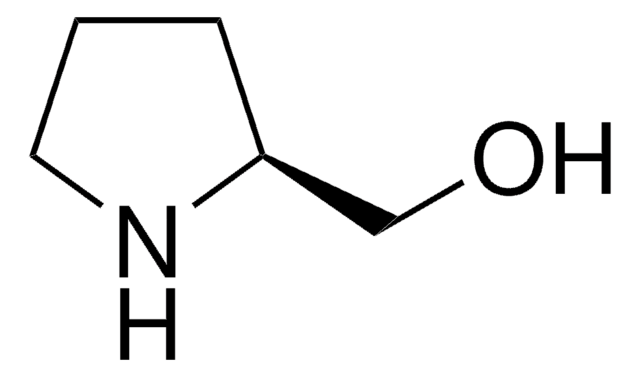
(S)-(+)-Pyrrolidine-3-carboxylic acid
≥98.0% (NT)
Synonym(s):
(S)-β-Proline
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H9NO2
CAS Number:
Molecular Weight:
115.13
Beilstein:
3537293
MDL number:
UNSPSC Code:
12352106
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥98.0% (NT)
form
solid
optical activity
[α]/D 20.0±2.0°, c = 2 in H2O
reaction suitability
reaction type: solution phase peptide synthesis
application(s)
peptide synthesis
storage temp.
2-8°C
SMILES string
OC(=O)[C@H]1CCNC1
InChI
1S/C5H9NO2/c7-5(8)4-1-2-6-3-4/h4,6H,1-3H2,(H,7,8)/t4-/m0/s1
InChI key
JAEIBKXSIXOLOL-BYPYZUCNSA-N
Related Categories
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
B R Huck et al.
Organic letters, 1(11), 1717-1720 (2000-06-03)
[formula: see text] Homooligomers of beta-amino acids (S)-3-pyrrolidine-3-carboxylic acid (PCA) and (S)-nipecotic acid (Nip) were studied by circular dichroism (CD) in methanol. In each series, a profound change in the far-UV CD spectrum was observed from monomer to tetramer, but
Alessandra Tolomelli et al.
Amino acids, 41(3), 575-586 (2011-05-05)
The synthesis of unusual cyclic amino acids, that may be envisaged as proline analogs, is an area of great interest for their potential applications as scaffolds for the design of bioactive peptidomimetics or units for the creation of novel foldamers.
Joanna Katarzyńska et al.
Journal of peptide science : an official publication of the European Peptide Society, 14(12), 1283-1294 (2008-09-30)
Immune response suppressors are used in the medical praxis to prevent graft rejection after organ transplantation and in the therapy of some autoimmune diseases. As a continuation of our previous work searching for new, effective suppressors devoid of toxicity, we
Antinociception by a peripherally administered novel endomorphin-1 analogue containing beta-proline.
Santi Spampinato et al.
European journal of pharmacology, 469(1-3), 89-95 (2003-06-05)
We previously described a novel endomorphin-1 analogue (Tyr-L-beta-Pro-Trp-Phe-NH(2); Endo1-beta-Pro) more resistant to enzymatic hydrolysis than endomorphin-1 that acts as a mu-opioid receptor agonist. In this study we report that Endo1-beta-Pro, s.c. injected in the mouse, is an effective antinociceptive agent
Cody Timmons et al.
The Journal of organic chemistry, 70(19), 7634-7639 (2005-09-10)
[reaction: see text] A new halo-Mannich-type reaction is reported using cyclopropyl carbonyl-derived enolates and sulfonyl-protected imines. Chiral oxazolidinones auxiliaries were found to be effective for completely controlling the stereochemistry of the products. Variations in the oxazolidinone, protecting group, and imine
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service