All Photos(2)
About This Item
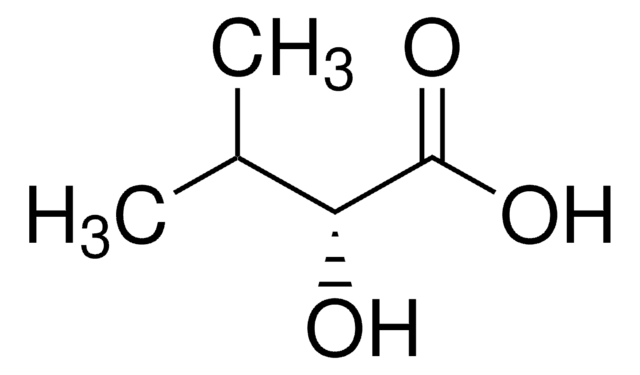
Empirical Formula (Hill Notation):
C4H8O3
CAS Number:
Molecular Weight:
104.10
Beilstein:
1720939
MDL number:
UNSPSC Code:
51113400
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥97.0% (T)
form
solid
optical purity
enantiomeric ratio: ≥99:1 (GC)
mp
50-54 °C
storage temp.
2-8°C
SMILES string
CC[C@@H](O)C(O)=O
InChI
1S/C4H8O3/c1-2-3(5)4(6)7/h3,5H,2H2,1H3,(H,6,7)/t3-/m1/s1
InChI key
AFENDNXGAFYKQO-GSVOUGTGSA-N
Other Notes
Chiral building block
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
M N Romanelli et al.
Chirality, 8(8), 579-584 (1996-01-01)
The enantiomers of 3-alpha-tropyl 2-(phenylthio)butyrate (SM32, 1) were prepared by chiral synthesis and tested for analgesic, cognition-enhancing, and ACh-releasing properties. They show enantioselectivity in some of the tests, the eutomer being related in configuration to R-(+)-hyoscyamine.
K.J. Hale et al.
Tetrahedron Letters, 36, 6965-6965 (1995)
M N Romanelli et al.
Chirality, 8(3), 225-233 (1996-01-01)
The enantiomers of two alpha-tropanyl esters, SM21 (1) and PG9 (2), derived from (+)-R-hyoscyamine, that act by increasing the central cholinergic tone, were obtained by esterification after resolution of the corresponding racemic acids [(-)-S-1, (-)-R-2 and (+)-S-2] and by stereospecific
Ferhan Siddiqi et al.
Biochemistry, 44(25), 9013-9021 (2005-06-22)
Mandelate racemase (MR, EC 5.1.2.2) from Pseudomonas putida catalyzes the Mg(2+)-dependent 1,1-proton transfer that interconverts the enantiomers of mandelate. Crystal structures of MR reveal that the phenyl group of all ground-state ligands is located within a hydrophobic cavity, remote from
R Guerranti et al.
Biochimica et biophysica acta, 1568(1), 45-52 (2001-12-04)
Rat liver L-threonine dehydrogenase is a mitochondrial enzyme which transforms L-threonine either into aminoacetone or into acetyl-CoA. We show that it is inhibited by several fatty acids and their derivatives: short chain fatty acids, L-2-hydroxybutyrate and D-3-hydroxybutyrate, long chain fatty
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


![[(3R)-3-Hydroxybutyryl]-L-carnitine analytical standard](/deepweb/assets/sigmaaldrich/product/structures/658/500/ff9570f8-a346-4077-9983-d0e67400bf47/640/ff9570f8-a346-4077-9983-d0e67400bf47.png)



![[(3R)-3-Hydroxyoctadecanoyl]-L-carnitine analytical standard](/deepweb/assets/sigmaaldrich/product/structures/195/646/9c581614-9449-4107-a05e-7c573a907483/640/9c581614-9449-4107-a05e-7c573a907483.png)