746991
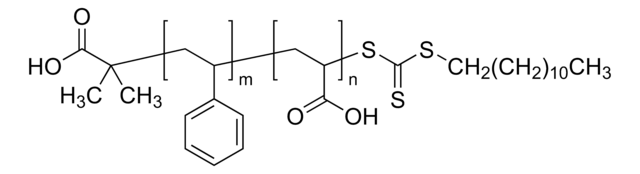
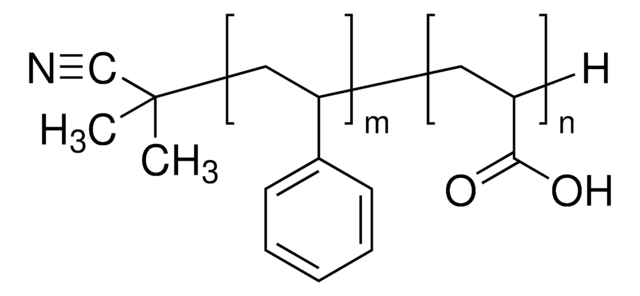
Polystyrene-block-poly(acrylic acid)
Synonym(s):
Poly(styrene)-block-poly(acrylic acid)
About This Item
Recommended Products
form
solid
mol wt
Mn 27,000-31,000 (polystyrene)
Mn 31,000-37,000 (total)
Mn 4,000-6,000 (poly(acrylic acid))
mp
258-263 °C
PDI
≤1.3
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
Legal Information
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Reversible addition–fragmentation chain transfer (RAFT) polymerization is rapidly moving to the forefront in construction of drug and gene delivery vehicles.
The development of drugs that target specific locations within the human body remains one of the greatest challenges in biomedicine today.
Over the past two decades, the rapid advance of controlled living polymerization (CLP) techniques.
Professor Robert K. Prud’homme introduces flash nanoprecipitation (FNP) for nanoparticle fabrication, which is a scalable, rapid mixing process for nanoparticle formulations.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service