Q4951
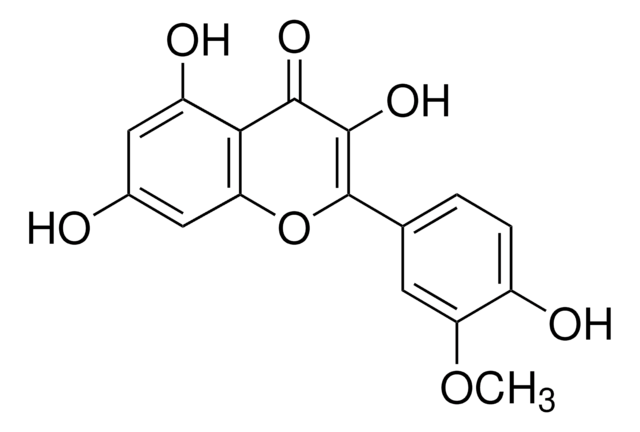
Quercetin
≥95% (HPLC), solid, anticancer agent
Synonyme(s) :
2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-4H-1-benzopyran-4-one, 3,3′,4′,5,6-Pentahydroxyflavone
About This Item
Produits recommandés
product name
Quercetin, ≥95% (HPLC), solid
Source biologique
synthetic (organic)
Pureté
≥95% (HPLC)
Forme
solid
Pf
316.5 °C
Solubilité
water: practically insoluble
Température de stockage
room temp
Chaîne SMILES
OC1=CC(O)=C2C(OC(C3=CC=C(O)C(O)=C3)=C(O)C2=O)=C1
InChI
1S/C15H10O7/c16-7-4-10(19)12-11(5-7)22-15(14(21)13(12)20)6-1-2-8(17)9(18)3-6/h1-5,16-19,21H
Clé InChI
REFJWTPEDVJJIY-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- Quercetin has been used as an antioxidant which reversed the immunosuppressive effects of high glucose and hyperglycemic sera in type 2 diabetic patients.
- It has been used as a detoxifying phytochemical in Apis mellifera.
- It has been used as a positive control in DPPH (2,2- diphenyl-1-picryhydrazyl) radical scavenging assay. It has also been used for the preparation of calibration curve to determine total flavonoid content.
Actions biochimiques/physiologiques
Caractéristiques et avantages
Notes préparatoires
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 3 Oral
Code de la classe de stockage
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe de danger pour l'eau (WGK)
WGK 3
Équipement de protection individuelle
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
Fatty acid synthesis supports cancer cell proliferation, essential for membrane generation, protein modification, and bioenergetics.
Discover Bioactive Small Molecules for Kinase Phosphatase Biology
Discover Bioactive Small Molecules for ADME/Tox
Discover Bioactive Small Molecules for Lipid Signaling Research
Protocoles
Protocol for HPLC Analysis of Flavonoids on Ascentis® RP-Amide
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique