796549
Stahl Aerobic Oxidation TEMPO solution
0.2 M in acetonitrile, Solution for Oxidation of Primary Alcohols
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Recommended Products
Quality Level
form
liquid
reaction suitability
reagent type: oxidant
concentration
0.2 M in acetonitrile
storage temp.
2-8°C
SMILES string
CN1C=CN=C1.CC2(C)CCCC(C)(C)N2[O].C3(C4=NC=CC=C4)=NC=CC=C3
InChI
1S/C10H8N2.C9H18NO.C4H6N2/c1-3-7-11-9(5-1)10-6-2-4-8-12-10;1-8(2)6-5-7-9(3,4)10(8)11;1-6-3-2-5-4-6/h1-8H;5-7H2,1-4H3;2-4H,1H3
InChI key
BQFURWVGIDXRNB-UHFFFAOYSA-N
General description
Stahl Aerobic Oxidation TEMPO solution contains 1-methylimidazole (NMI), 2,2′-bipyridyl (bpy), and TEMPO. It is widely employed for the oxidation of alcohols.
Application
This convenience solution can be used in tandem with tetrakisacetonitrile copper(I) triflate (685038) for the oxidation of primary alcohols through aerobic conditions developed by the Stahl Research Group.
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Dam. 1 - Flam. Liq. 2 - Repr. 2 - Skin Corr. 1C
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
35.6 °F
Flash Point(C)
2.0 °C
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Continuous-Flow Aerobic Oxidation of Primary Alcohols with a Copper(I)/TEMPO Catalyst
Greene, JF, et al.
Organic Process Research & Development, 17 (10), 1247?1251-1247?1251 (2013)
Practical Aerobic Alcohol Oxidation with Cu/Nitroxyl and Nitroxyl/NOx Catalyst Systems.
Miles KC and Stahl SS.
Aldrichimica Acta, 48(1) (2015)
Jessica M Hoover et al.
Journal of the American Chemical Society, 133(42), 16901-16910 (2011-08-25)
Aerobic oxidation reactions have been the focus of considerable attention, but their use in mainstream organic chemistry has been constrained by limitations in their synthetic scope and by practical factors, such as the use of pure O(2) as the oxidant
Janelle E Steves et al.
The Journal of organic chemistry, 80(21), 11184-11188 (2015-10-13)
Two solutions, one consisting of bpy/TEMPO/NMI and the other bpy/ABNO/NMI (bpy =2,2'-bipyridyl; TEMPO = 2,2,6,6-tetramethylpiperidine N-oxyl, ABNO = 9-azabicyclo[3.3.1]nonane N-oxyl; NMI = N-methylimidazole), in acetonitrile are shown to have good long-term stability (≥1 year) under air at 5 °C. The
Jessica M Hoover et al.
Nature protocols, 7(6), 1161-1166 (2012-05-29)
This protocol describes a practical laboratory-scale method for aerobic oxidation of primary alcohols to aldehydes, using a chemoselective Cu(I)/TEMPO (TEMPO = 2,2,6,6-tetramethyl-1-piperidinyloxyl) catalyst system. The catalyst is prepared in situ from commercially available reagents, and the reactions are performed in
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
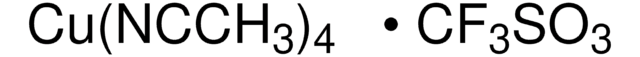
![9-Azabicyclo[3.3.1]nonane N-oxyl 95%](/deepweb/assets/sigmaaldrich/product/structures/287/155/e2f4a2e1-1d4e-4bed-9187-9e16d23cbbbf/640/e2f4a2e1-1d4e-4bed-9187-9e16d23cbbbf.png)



![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)









