15404
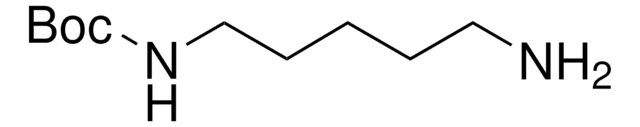
N-Boc-1,4-butanediamine
≥97.0% (GC/NT)
Synonym(s):
N-Boc-1,4-diaminobutane, tert-Butyl N-(4-aminobutyl)carbamate
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(CH3)3COCONH(CH2)4NH2
CAS Number:
Molecular Weight:
188.27
Beilstein:
1937878
MDL number:
UNSPSC Code:
12352116
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥97.0% (GC/NT)
reaction suitability
reagent type: cross-linking reagent
refractive index
n20/D 1.460
density
0.984 g/mL at 20 °C (lit.)
functional group
Boc
amine
SMILES string
NCCCCNC(OC(C)(C)C)=O
InChI
1S/C9H20N2O2/c1-9(2,3)13-8(12)11-7-5-4-6-10/h4-7,10H2,1-3H3,(H,11,12)
InChI key
ZFQWJXFJJZUVPI-UHFFFAOYSA-N
Application
- Carboxy-Silane Coated Iron Oxide Nanoparticles: Details the application of N-Boc-1,4-butanediamine in modifying iron oxide nanoparticles for imaging and drug delivery (D Stanicki, S Boutry, S Laurent, et al., 2014). Access the article.
Other Notes
Preparation of pharmacologically active compounds. Preparation of spermidine analogues. Introduction of a C4-spacer.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
228.2 °F - closed cup
Flash Point(C)
109.0 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Y Shai et al.
Biochemistry, 28(11), 4801-4806 (1989-05-30)
In the present study we synthesize 18F-labeled insulin of high specific radioactivity. A new prosthetic group methodology, in which [18F]fluoride displaces a bromide group of 4-(bromomethyl)-benzoylamine intermediates, was used. The 4-(fluoromethyl)benzoyl product was chemically stable. 18F-Labeled insulin retains the essential
Regina Holm et al.
Macromolecular rapid communications, 36(23), 2083-2091 (2015-09-08)
In this work, the synthesis of polypeptoid-block-polypeptide copolymers (block copolypept(o)ides) based on bifunctional initiators is described, which introduces a distinct chemical entity at the connection between both blocks. With a view towards redox-sensitive block copolypept(o)ides, a cystamine-based initiator was used
Hongyan Guo et al.
Journal of medicinal chemistry, 45(10), 2056-2063 (2002-05-03)
Several iron chelators containing alpha,beta-unsaturated hydroxamic acid motifs appended to a citric acid platform were synthesized. Mycobacterium paratuberculosis was then challenged to grow in the presence of a panel of siderophores (mycobactin J, deferrioxamine B, acinetoferrin, and nannochelin A) and
M. McWatt, G.J. Boons et al.
European Journal of Organic Chemistry, 2535-2535 (2001)
Abshar Hasan et al.
Frontiers in chemistry, 8, 416-416 (2020-06-13)
Poly(N-substituted glycine) "peptoids" are an interesting class of peptidomimics that can resist proteolysis and mimic naturally found antimicrobial peptides (AMPs), which exhibit wide spectrum activity against bacteria. This work investigates the possibility of modifying peptoid AMP mimics (AMPMs) with aliphatic
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service