153818
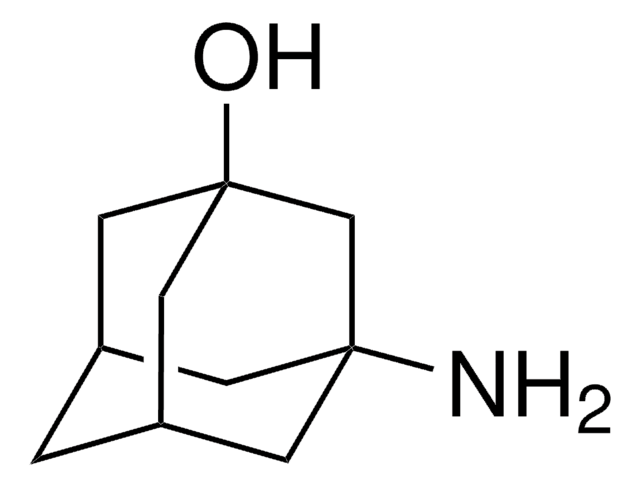
2-Adamantylamine hydrochloride
99%
Synonym(s):
2-Adamantanamine hydrochloride, 2-Aminoadamantane hydrochloride
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C10H17N · HCl
CAS Number:
Molecular Weight:
187.71
Beilstein:
4297901
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
solid
mp
>300 °C (lit.)
SMILES string
Cl.NC1[C@@H]2C[C@H]3C[C@@H](C2)C[C@@H]1C3
InChI
1S/C10H17N.ClH/c11-10-8-2-6-1-7(4-8)5-9(10)3-6;/h6-10H,1-5,11H2;1H/t6-,7+,8-,9+,10?;
InChI key
WLDWDRZITJEWRJ-ZDAMNCSYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
The HPLC assay of 2-adamantylamine hydrochloride after pre-column derivatization with 4-fluoro-7-nitro-2,1,3-benzoxadiazole has been studied.
Application
2-Adamantylamine hydrochloride was used to prepare 2-adamantylamide of 2′-(carboxymethoxime)-olivomycin I.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
S V Krapivin et al.
Biulleten' eksperimental'noi biologii i meditsiny, 116(11), 515-518 (1993-11-01)
The action of the new stimulant bromantane on spectra power EEG on Fourier of sensorimotor cortex, dorsal hippocamp and lateral hypothalamus of left and right hemispheres of brain of rat in free behavior was investigated. Bromantane leads to decreases in
Yasuhiko Higashi et al.
Biomedical chromatography : BMC, 20(5), 423-428 (2005-09-15)
Simultaneous HPLC assay of 1-adamantanamine hydrochloride (amantadine) and its four related compounds [2-adamantanamine hydrochloride (2-ADA), 1-adamantanmethylamine (ADAMA), 1-(1-adamantyl)ethylamine hydrochloride (rimantadine) and 3,5-dimethyl-1-adamantanamine hydrochloride (memantine)] in phosphate-buffered saline (pH 7.4) after pre-column derivatization with 4-fluoro-7-nitro-2,1,3-benzoxadiazole (NBD-F) was developed. Phosphate-buffered saline samples
Anna N Tevyashova et al.
The Journal of antibiotics, 62(1), 37-41 (2009-01-10)
A novel way of chemical modification of the antibiotic olivomycin I at the 2'-keto group of the side chain of the aglycone moiety was developed. Reaction of olivomycin I with the carboxymethoxylamine hemihydrochloride gave the key intermediate, 2'-carboxymethoxime-olivomycin I, which
C Clark et al.
Immunopharmacology, 21(1), 41-50 (1991-01-01)
The present in vitro investigations on amantadine (AmTd) and its isomer 2-aminoadamantane (2-NH2-Adam), and the corresponding analogs, 1-nitroadamantane (1-NO2-Adam) and 2-nitroadamantane (2-NO2-Adam), were undertaken to gain information about molecular features that might have a dominant role in inhibiting T lymphocyte
Iu V Vakhitova et al.
Molekuliarnaia biologiia, 39(2), 276-285 (2005-04-29)
We have used the Rat Atlas cDNA Array ("BD Bioscience") to assess changes in mRNA expression of 588 genes in rat brain after acute treatment of 2-aminoadamantane compound--Ladasten. Drug exhibits the psychostimulating and anxyolitic actions. The analysis of results of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service









![1-Bicyclo[1.1.1]pentylamine hydrochloride](/deepweb/assets/sigmaaldrich/product/structures/287/052/55f4f60a-a9e0-4ea2-b1e8-5b3f6ce0ff21/640/55f4f60a-a9e0-4ea2-b1e8-5b3f6ce0ff21.png)

