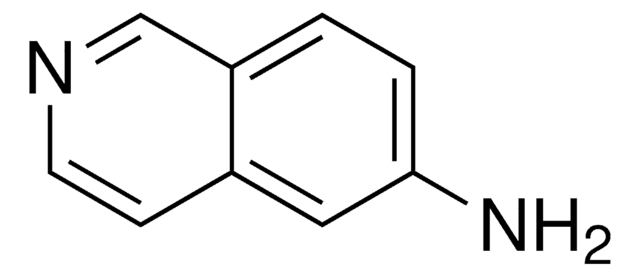
A79205
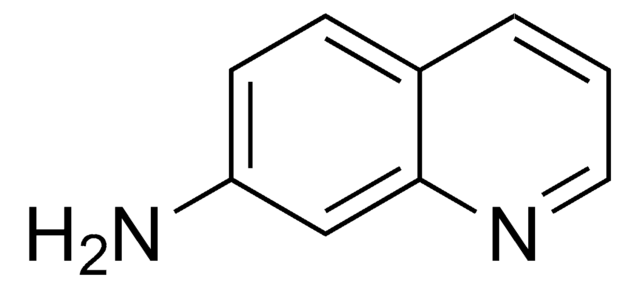
5-Aminoquinoline
97%
Synonym(s):
5-Quinolinamine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C9H8N2
CAS Number:
Molecular Weight:
144.17
Beilstein:
114479
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
powder
bp
310 °C (lit.)
mp
106-109 °C (lit.)
SMILES string
Nc1cccc2ncccc12
InChI
1S/C9H8N2/c10-8-4-1-5-9-7(8)3-2-6-11-9/h1-6H,10H2
InChI key
XMIAFAKRAAMSGX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
V Arjunan et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 74(5), 1215-1223 (2009-10-27)
The Fourier transform infrared (FTIR) and FT-Raman spectra of 5-aminoquinoline (5AQ) have been recorded in the range 4000-400 and 3500-100 cm(-1), respectively. The complete vibrational assignment and analysis of the fundamental modes of the compounds were carried out using the
R Banoo et al.
Indian journal of biochemistry & biophysics, 30(4), 229-233 (1993-08-01)
In vitro oxidation and acetylation of 5-aminoquinoline by rabbit liver enzyme preparation has been investigated. Incubation of 5-aminoquinoline with cytosol fraction of the enzyme preparation in the presence of acetyl coenzyme-A gave rise to three different products, viz. 5-amino-2-hydroxy quinoline
Jagat P Bridhkoti et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 79(3), 412-417 (2011-04-20)
Photophysical properties of 5-aminoquinoline (5AQ) have been investigated in various non-polar and polar (protic and aprotic) solvents using steady state and time resolved fluorescence. In aprotic solvents, the spectral maxima depend on the polarity. However, in protic solvents both the
B G Siim et al.
Biochemical pharmacology, 48(8), 1593-1604 (1994-10-18)
The 4-alkylamino-5-nitroquinolines (5NQs) are a new series of bioreductive drugs that exhibit varying degrees of selective toxicity (up to 60-fold) under hypoxic conditions in cell culture. This study tested the hypothesis that differences in hypoxia-selective cytotoxicity in this series reflect
Maria Sittig et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 27(3), 1072-1079 (2020-09-29)
Quinoline photobases exhibit a distinctly higher pKa in their electronically excited state than in the ground state, thereby enabling light-controlled proton transfer reactions, for example, in molecular catalysis. The absorption of UV light translates to a pKa jump of approximately
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service