B84904
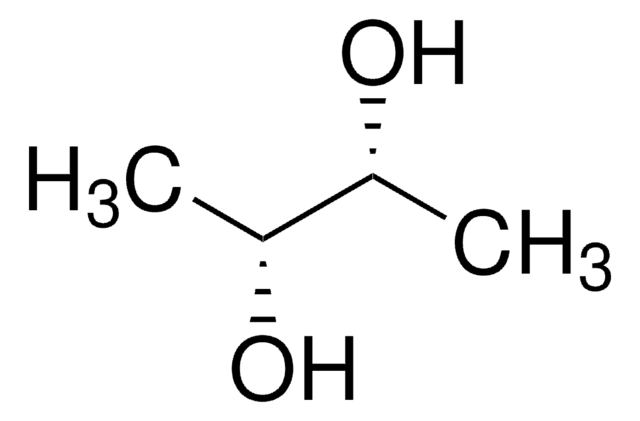
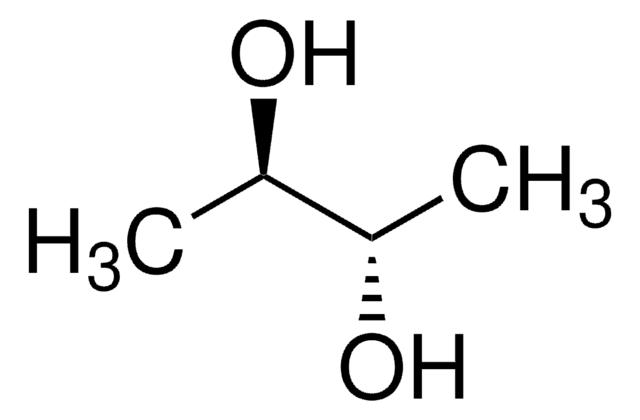
2,3-Butanediol
98%
Synonym(s):
2,3-Butylene glycol
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3CH(OH)CH(OH)CH3
CAS Number:
Molecular Weight:
90.12
Beilstein:
969165
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
liquid
refractive index
n20/D 1.433 (lit.)
bp
183-184 °C (lit.)
mp
25 °C (lit.)
density
1.002 g/mL at 20 °C (lit.)
SMILES string
CC(O)C(C)O
InChI
1S/C4H10O2/c1-3(5)4(2)6/h3-6H,1-2H3
InChI key
OWBTYPJTUOEWEK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2,3-Butanediol (2,3-BDO) is used as precursor in the synthesis of cyclic carbonates and methyl ethyl ketone.
Application
- CRISPR/Cas9 in Pichia pastoris: 2,3-Butanediol (BDO) is used in the construction of P. pastoris cell factories as a representative example to demonstrate the procedures for integrating multiple heterologous genes using the CRISPR-based multiplex genome integration toolkit (Gao et al., 2024).
- Antifungal activity in sorghum: 2,3-butanediol, a metabolite of A. oryzae YRA3, can induce the production of root exudates which modulate the growth of the rhizospheric fungi and bacteria (Rashad et al., 2023).
Storage Class Code
10 - Combustible liquids
WGK
WGK 1
Flash Point(F)
185.0 °F - closed cup
Flash Point(C)
85 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Sustainable Synthesis of Non-Isocyanate Polyurethanes Based on Renewable 2, 3-Butanediol
A Kirchberg, et al.
Macromolecular Chemistry and Physics, 223, 2200010-2200010 (2022)
Natsuo Ueda et al.
Neuropharmacology, 48(8), 1079-1085 (2005-05-25)
It is widely accepted that fatty acid amide hydrolase (FAAH) plays a central role in the hydrolysis of anandamide. However, we found a second N-acylethanolamine hydrolase in animal tissues which hydrolyzed anandamide at acidic pH. This "acid amidase" was first
E Celińska et al.
Biotechnology advances, 27(6), 715-725 (2009-05-16)
Biotechnological production of 2,3-butanediol (hereafter referred to as 2,3-BD) from wastes and excessive biomass is a promising and attractive alternative for traditional chemical synthesis. In the face of scarcity of fossil fuel supplies the bio-based process is receiving a significant
Patricia Hermand et al.
The Journal of biological chemistry, 278(7), 4892-4898 (2002-12-13)
ICAM-4 (LW blood group glycoprotein) is an erythroid-specific membrane component that belongs to the family of intercellular adhesion molecules and interacts in vitro with different members of the integrin family, suggesting a potential role in adhesion or cell interaction events
Frank Plaisant et al.
Free radical biology & medicine, 34(7), 862-872 (2003-03-26)
The pathophysiology of brain lesions associated with cerebral palsy is multifactorial and likely involves excess release of glutamate and excess production of free radicals, among other factors. Theoretically, antioxidants could limit the severity of these brain lesions. Peroxiredoxins are a
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service