N33101
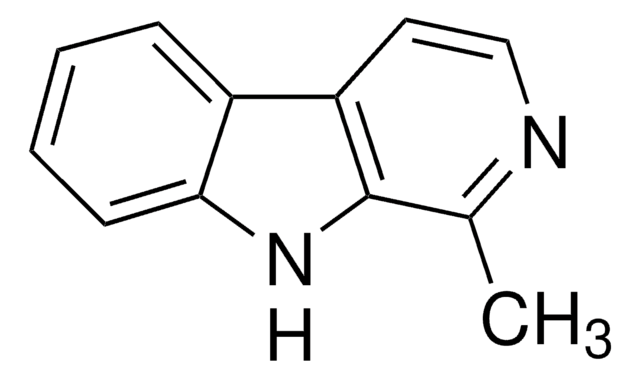
Norharmane
98%
Synonym(s):
β-Carboline, 9H-Pyrido[3,4-b]indole
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C11H8N2
CAS Number:
Molecular Weight:
168.19
MDL number:
UNSPSC Code:
12352202
PubChem Substance ID:
Recommended Products
Assay
98%
SMILES string
c1ccc2c(c1)[nH]c3cnccc23
InChI
1S/C11H8N2/c1-2-4-10-8(3-1)9-5-6-12-7-11(9)13-10/h1-7,13H
InChI key
AIFRHYZBTHREPW-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Biochem/physiol Actions
Inhibitor of indoleamine 2,3-dioxygenase (IDO).
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Rosa Busquets et al.
Molecular nutrition & food research, 53(12), 1496-1504 (2009-10-08)
Heterocyclic amines (HCAs) are potent mutagens/carcinogens to which humans are frequently exposed through the consumption of cooked meat and fish food. The effect of normal intake of HCAs and their role in the aetiology of human cancer is unknown. To
Olga I Tarzi et al.
Journal of mass spectrometry : JMS, 44(2), 260-277 (2008-11-18)
The thermal stability of several commonly used crystalline matrix-assisted ultraviolet laser desorption/ionization mass spectrometry (UV-MALDI-MS) matrices, 2,5-dihydroxybenzoic acid (gentisic acid; GA), 2,4,6-trihydroxyacetophenone (THA), alpha-cyano-4-hydroxycinnamic acid (CHC), 3,5-dimethoxy-4-hydroxycinnamic acid (sinapinic acid; SA), 9H-pirido[3,4-b]indole (nor-harmane; nor-Ho), 1-methyl-9H-pirido[3,4-b]indole (harmane; Ho), perchlorate of nor-harmanonium
Andrés G León et al.
Journal of chromatography. A, 1192(2), 254-258 (2008-04-25)
The presence of cyclodextrins (CDs) in the mobile phase alters the chromatographic equilibria and induces a secondary chemical equilibrium associated to the chromatographic separation by HPLC. In this study the influence of the presence of CDs in the mobile phase
M Micaela Gonzalez et al.
Photochemical & photobiological sciences : Official journal of the European Photochemistry Association and the European Society for Photobiology, 8(8), 1139-1149 (2009-07-30)
The photochemistry of norharmane (9H-pyrido[3,4-b]indole) in acidic (pH 5.0+/-0.1) and alkaline (pH 10.0+/-0.1) aqueous solutions was studied. The photochemical reactions were monitored by TLC, UV/VIS absorption spectroscopy, high-performance liquid chromatography (HPLC), electronic ionization-mass spectrometry (EI-MS), UV-laser desorption/ionization-time of flight-mass spectrometry
D Reyman et al.
Magnetic resonance in chemistry : MRC, 45(10), 830-834 (2007-08-31)
In this work, we have analysed the tendency of two beta-carboline derivatives, harmane and norharmane, in the formation of hydrogen bonds. We obtained the (1)H and (13)C NMR spectra of different mixtures of these derivatives with acetic acid (AcOH) in
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![2-Amino-9H-pyrido[2-3-b]indole ≥98% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/351/152/37c98523-590f-4b27-826c-5b3d4b502047/640/37c98523-590f-4b27-826c-5b3d4b502047.png)
![1,2,3,4-Tetrahydro-9H-pyrido[3,4-b]indole 98%](/deepweb/assets/sigmaaldrich/product/structures/181/460/3d58bc34-1b5c-4295-bbac-3b52085670e8/640/3d58bc34-1b5c-4295-bbac-3b52085670e8.png)