535087
2-Fluoroadenine
96%
Synonym(s):
2-Fluoro-7(9)H-purin-6-ylamine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H4FN5
CAS Number:
Molecular Weight:
153.12
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
96%
mp
>350 °C (lit.)
functional group
fluoro
SMILES string
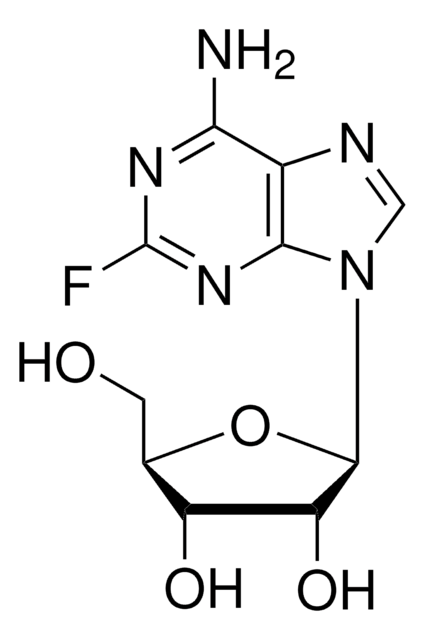
Nc1[nH]c(F)nc2ncnc12
InChI
1S/C5H4FN5/c6-5-10-3(7)2-4(11-5)9-1-8-2/h1H,(H3,7,8,9,10,11)
InChI key
WKMPTBDYDNUJLF-UHFFFAOYSA-N
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Sepideh Afshar et al.
Protein science : a publication of the Protein Society, 18(5), 1107-1114 (2009-04-24)
A double mutant of human purine nucleoside phosphorylase (hDM) with the amino acid mutations Glu201Gln:Asn243Asp cleaves adenosine-based prodrugs to their corresponding cytotoxic drugs. When fused to an anti-tumor targeting component, hDM is targeted to tumor cells, where it effectively catalyzes
Song Ye et al.
Nucleosides, nucleotides & nucleic acids, 22(10), 1899-1905 (2003-11-12)
A convenient synthesis of 2'-deoxy-2-fluoroadenosine from commercially available 2-fluoroadenine is described. The coupling reaction of silylated 2-fluoroadenine with phenyl 3,5-bis[O-(t-butyldimethylsilyl)]-2-deoxy-1-thio-D-erythro-pentofuranoside gave the corresponding 2-fluoro-2'-deoxyadenosine derivative (alpha/beta = 1:1) in good yield. The alpha- and beta-anomers were separated by chromatography, and
X Y Wang et al.
Gene therapy, 11(21), 1559-1567 (2004-09-03)
Gene-directed enzyme prodrug therapy (GDEPT) based on the Escherichia coli enzyme, purine nucleoside phosphorylase (PNP), provides a novel strategy for treating slowly growing tumors like prostate cancer (CaP). PNP converts systemically administered prodrug, fludarabine phosphate, to a toxic metabolite, 2-fluoroadenine
B E Miller et al.
Cancer research, 46(1), 89-93 (1986-01-01)
A series of subpopulation lines derived from a single mouse mammary tumor were tested for their ability to interact with each other in metabolic cooperation assays in three-dimensional collagen gel cultures. Inhibition of growth in the presence of selective drug
Bénédicte Trouiller et al.
Biotechnology letters, 29(10), 1591-1598 (2007-06-15)
The moss, Physcomitrella patens, is a novel tool in plant functional genomics due to its exceptionally high gene targeting efficiency that is so far unique for plants. To determine if this high gene targeting efficiency is exclusive to P. patens
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service