128090
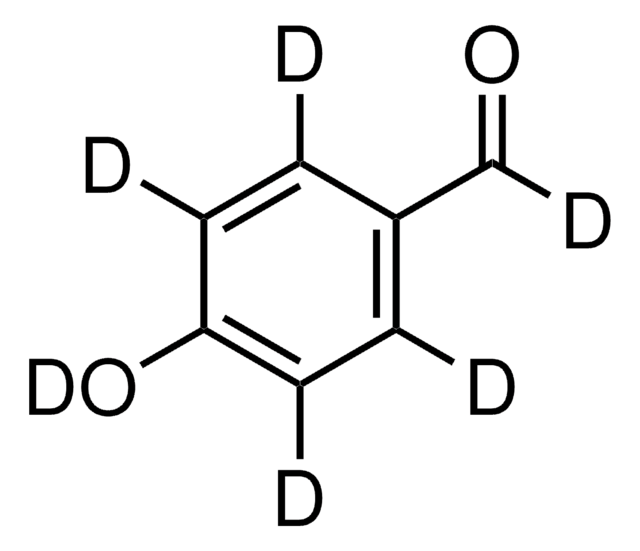
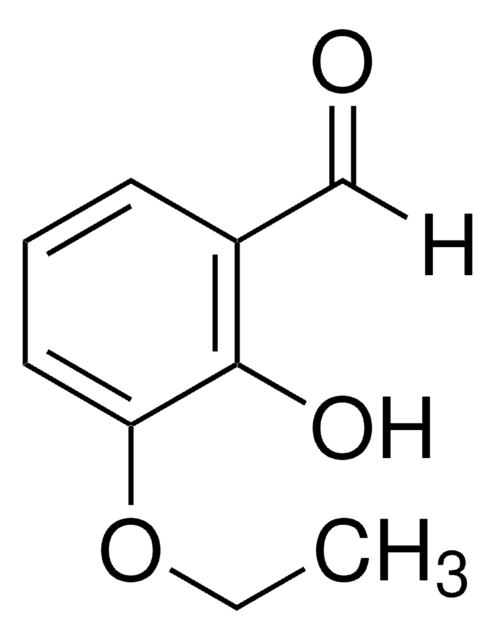
3-Ethoxy-4-hydroxybenzaldehyde
ReagentPlus®, 99%
Synonym(s):
Ethylvanillin, NSC 1803, NSC 67240
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
C2H5OC6H3(OH)CHO
CAS Number:
Molecular Weight:
166.17
Beilstein:
1073761
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
vapor pressure
<0.01 mmHg ( 25 °C)
Quality Level
product line
ReagentPlus®
Assay
99%
mp
74-77 °C (lit.)
functional group
aldehyde
SMILES string
[H]C(=O)c1ccc(O)c(OCC)c1
InChI
1S/C9H10O3/c1-2-12-9-5-7(6-10)3-4-8(9)11/h3-6,11H,2H2,1H3
InChI key
CBOQJANXLMLOSS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Ethylvanillin (3-Ethoxy-4-hydroxybenzaldehyde) is an artificial additive for improving taste. It chemiluminesces by reacting with potassium permanganate in sulphuric or polyphosphoric acid media.
Application
Ethylvanillin (3-Ethoxy-4-hydroxybenzaldehyde) was used as an internal standard in the determination of phenolic compounds obtained from (alkaline) CuO-based oxidation of humic acid.
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
293.0 °F - closed cup
Flash Point(C)
145 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Diana L D Lima et al.
Talanta, 72(4), 1404-1409 (2007-06-15)
Operational parameters like migration time, temperature, voltage, composition of background electrolyte and content of organic modifier were optimized in CZE for the determination of lignin-like phenolic compounds. The applied background electrolyte buffer consisted of a Na(2)B(4)O(7), KH(2)PO(4) aqueous solution, pH
Meropi Timotheou-Potamia et al.
Talanta, 71(1), 208-212 (2007-01-15)
Vanillin, ethylvanillin and 4-hydroxy-3-methoxy-benzylalcohol have been found to chemiluminesce by the action of potassium permanganate in sulphuric or polyphosphoric acid media. Both acid media have been compared and sulphuric acid allows the sensitive determination of 0.15-10.0, 0.010-1.0 and 0.0030-0.30microg mL(-1)
David Kahoun et al.
Journal of chromatography. A, 1202(1), 19-33 (2008-07-16)
The objective of this study was the determination of 25 phenolic compounds in different mead samples (honeywines) using high performance liquid chromatography (HPLC) with coulometric-array detection and in case of hydroxymethylfurfural with UV detection. Our method was optimized in respect
E Jagerdeo et al.
Journal of AOAC International, 83(1), 237-240 (2000-02-29)
This paper describes a reversed-phase liquid chromatographic method for the determination of vanillin, associated natural aromatic compounds and/or synthetic precursors, ethyl vanillin, and coumarin, a commonly encountered adulterant in nonbeverage and beverage alcohol products using a ternary gradient mobile phase.
Hyun-Joo Jung et al.
Archives of pharmacal research, 33(2), 309-316 (2010-03-03)
The present work aimed to assess novel pharmacological properties of ethyl vanillin (EVA) which is used as a flavoring agent for cakes, dessert, confectionary, etc. EVA exhibited an inhibitory activity in the chorioallantoic membrane angiogenesis. Anti-inflammatory activity of EVA was
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![3-ETHOXY-2-HYDROXYBENZALDEHYDE (6-TERT-PENTYL-5,6,7,8-TETRAHYDRO[1]BENZOTHIENO[2,3-D]PYRIMIDIN-4-YL)HYDRAZONE AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/332/205/e4209595-7039-435f-a637-38bacf5fe1c3/640/e4209595-7039-435f-a637-38bacf5fe1c3.png)