H4405
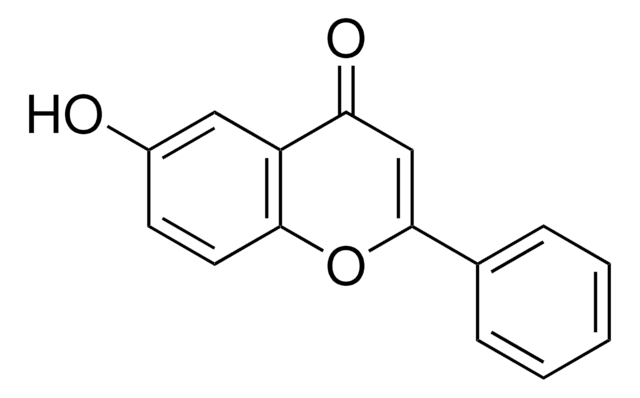
5-Hydroxyflavone
≥97%
Sinónimos:
5-Hydroxy-2-phenylchromone, NSC 26745, Primuletin
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C15H10O3
Número de CAS:
Peso molecular:
238.24
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Ensayo
≥97%
cadena SMILES
Oc1cccc2OC(=CC(=O)c12)c3ccccc3
InChI
1S/C15H10O3/c16-11-7-4-8-13-15(11)12(17)9-14(18-13)10-5-2-1-3-6-10/h1-9,16H
Clave InChI
IYBLVRRCNVHZQJ-UHFFFAOYSA-N
Información sobre el gen
rat ... Gabra2(29706)
Aplicación
Reactant involved in:
- Condensation reactions for synthesis of copper(II) complexes as bioactive molecules to combat antioxidants
- Thermal behavior studies of vanadyl complexes with flavone derivatives in terms of insulin-mimetic agents
- O-methylation with di-Me carbonate
- DFT studies on excited-state intramolecular proton transfer
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Errol G Lewars et al.
Rapid communications in mass spectrometry : RCM, 21(10), 1669-1679 (2007-04-28)
In a recent study of the collision-induced dissociation of protonated and deprotonated molecules of 3-, 5-, 6-, 7-, 2'-, 3'- and 4'-hydroxyflavone, it was observed that the ratio, gamma, of the propensities for cross-ring cleavage (CRC) to ring opening (RO)
A R Ibrahim et al.
Applied and environmental microbiology, 55(12), 3140-3142 (1989-12-01)
The conversion of 5-hydroxyflavone by various microorganisms was studied. Among them, Streptomyces fulvissimus was the sole microbe which produced a new polar metabolite from 5-hydroxyflavone in addition to 5,4-dihydoxy- and 5,3,4-trihydroxyflavone. The structure of this polar metabolite was determined to
Sankarlal Ash et al.
Journal of molecular modeling, 16(5), 831-839 (2009-09-18)
Potential energy (PE) curves for the intramolecular proton transfer in the ground (GSIPT) and excited (ESIPT) states of 3-hydroxy-flavone (3HF) and 5-hydroxy-flavone (5HF) were studied using DFT/B3LYP (6-31G (d,p)) and TD-DFT/B3LYP (6-31G (d,p)) level of theory respectively. Our calculations suggest
R Sai Sathish et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 69(1), 282-285 (2007-05-29)
In the presence of Al(III) ions, 5-hydroxyflavone (5HF) through a complexation reaction in MeOH, shows dual fluorescence, characterized by a newly developed peak at 554nm upon excitation at 363nm. In this communication, the subsequent ligand exchange reaction of the complex
Wimal Herath et al.
Chemical & pharmaceutical bulletin, 56(4), 418-422 (2008-04-02)
5,7-Dihydroxyflavone (chrysin) (1) when fermented with fungal cultures, Aspergillus alliaceous (ATCC 10060), Beauveria bassiana (ATCC 13144) and Absidia glauco (ATCC 22752) gave mainly 4'-hydroxychrysin (4), chrysin 7-O-beta-D-4-O-methylglucopyranoside (5) and chrysin 7-sulfate (6), respectively. Mucore ramannianus (ATCC 9628), however, transformed chrysin
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico