55520
3-Hydroxymandelic acid
≥97.0% (T)
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C8H8O4
Número de CAS:
Peso molecular:
168.15
Beilstein:
2365378
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Análisis
≥97.0% (T)
mp
128-132 °C
grupo funcional
carboxylic acid
hydroxyl
cadena SMILES
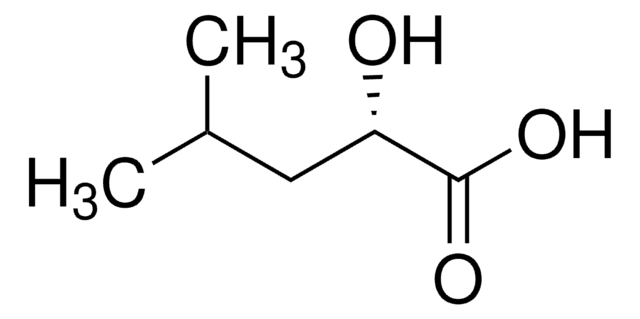
OC(C(O)=O)c1cccc(O)c1
InChI
1S/C8H8O4/c9-6-3-1-2-5(4-6)7(10)8(11)12/h1-4,7,9-10H,(H,11,12)
Clave InChI
OLSDAJRAVOVKLG-UHFFFAOYSA-N
Descripción general
3-Hydroxymandelic acid is a hydroxy acid derivative. Chiral separation of 3-hydroxymandelic acid has been achieved by ligand-exchange capillary electrochromatography.
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
J M Midgley et al.
Biomedical mass spectrometry, 6(11), 485-490 (1979-11-01)
o-Hydroxymandelic acid and m-hydroxymandelic acid have been identified in human urine by gas chromatography mass spectrometry selected ion monitoring. After solvent extraction the urinary acids were converted to their O-trifluoroacetoxy methyl ester derivatives which were identified by comparison of the
K E Ibrahim et al.
The Journal of pharmacy and pharmacology, 35(3), 144-147 (1983-03-01)
The metabolism of R-(-)-m-synephrine (administered orally and by inhalation in man and intraperitoneally in rats) was studied quantitatively by a gas chromatography-mass spectrometry-selected ion monitoring (g.c.-m.s.-s.i.m.) method using deuterated internal standards. When m-synephrine hydrochloride was administered orally to humans in
M W Couch et al.
Clinica chimica acta; international journal of clinical chemistry, 158(1), 109-114 (1986-07-15)
The urinary concentrations of o-hydroxymandelic acid, m-hydroxymandelic acid, p-hydroxymandelic acid, homovanillic acid and vanillylmandelic acid were determined in 57 healthy children and 9 patients with neuroblastoma. The concentrations of o-hydroxymandelic acid and p-hydroxymandelic were not significantly different for both groups
Role of the charge in continuous beds in the chiral separation of hydroxy acids by ligand-exchange capillary electrochromatography.
Lecnik O, et al.
Electrophoresis, 24(17), 2983-2985 (2003)
K Gumbhir et al.
Journal of pharmaceutical and biomedical analysis, 12(7), 943-949 (1994-07-01)
An LC method for the analysis of m-hydroxymandelic acid (MHMA) and m-hydroxyphenylglycol (MHPG) and their conjugates in human plasma was developed and validated. The method for the quantitation involved extraction of acidified plasma (subject to hydrolysis with beta-glucuronidase for 120
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico