Select a Size
| Size/SKU | Availability | Price |
|---|---|---|
10 μg | Please contact Customer Service for Availability | €595.00 |
€595.00
recombinant
expressed in mouse NSO cells
Quality Segment
assay
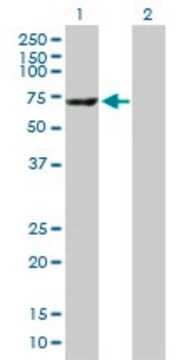
≥95% (SDS-PAGE)
form
solution
specific activity
≥1.0 EU/μg, 30,000 pmol/min-μg protein
mol wt
predicted mol wt ~60 kDa
concentration
0.4-0.6 mg/mL
impurities
≤1.0 EU/μg endotoxin
shipped in
dry ice
storage temp.
−70°C
Gene Information
human ... CES2(8824)
Biochem/physiol Actions
Physical form
Other Notes
1 of 1
This Item | |||
|---|---|---|---|
| Gene Information human ... CES2(8824) | Gene Information human ... CES2(8824) | Gene Information - | Gene Information human ... CES1(1066) |
| assay ≥95% (SDS-PAGE) | assay - | assay - | assay - |
| specific activity ≥1.0 EU/μg, 30,000 pmol/min-μg protein | specific activity - | specific activity ≥500 units/mg protein | specific activity - |
| concentration 0.4-0.6 mg/mL | concentration - | concentration - | concentration - |
| form solution | form buffered aqueous solution | form liquid | form buffered aqueous solution |
| recombinant expressed in mouse NSO cells | recombinant - | recombinant expressed in baculovirus infected BTI insect cells | recombinant - |
Still not finding the right product?
Explore all of our products under Carboxylesterase 2 human
signalword
Danger
hcodes
pcodes
Hazard Classifications
Resp. Sens. 1
Storage Class
10 - Combustible liquids
wgk
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.