550809
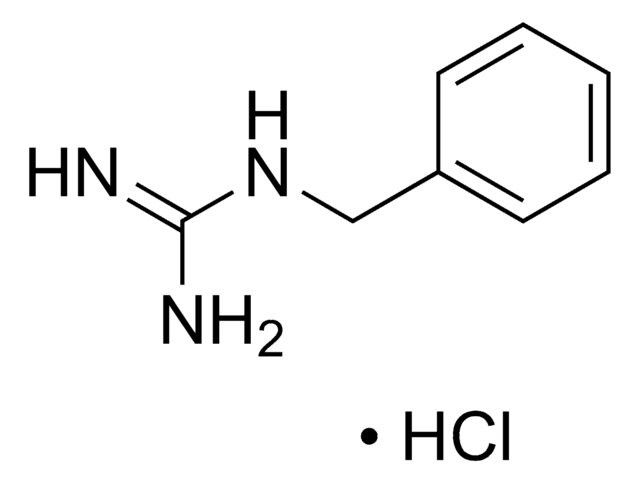
Phenylguanidine carbonate salt
≥99%
Synonym(s):
1-Phenylguanidine carbonate, N-Phenylguanidine carbonate
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
C6H5NHC(=NH)NH2·(H2CO3)x
CAS Number:
Molecular Weight:
135.17
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥99%
mp
149-153 °C (lit.)
SMILES string
OC(O)=O.NC(=N)Nc1ccccc1
InChI
1S/C7H9N3.CH2O3/c8-7(9)10-6-4-2-1-3-5-6;2-1(3)4/h1-5H,(H4,8,9,10);(H2,2,3,4)
InChI key
XDSYAIICRRZSJX-UHFFFAOYSA-N
Application
Phenylguanidine carbonate salt may be used in the preparation of the following bioactive compounds:
- (E)-methyl 2-(2-((6-cyclopropyl-2-(phenylamino)pyrimidin-4-yloxy)methyl)phenyl)-3-methoxyacrylate
- cyprodinil
- 5,6-dihydropyrido[2,3-d]pyrimidine
- ethyl 1-methyl-8-(phenylamino)-4,5-dihydro-1H-pyrazolo[4,3-h]quinazoline-3-carboxylate
- 2-(phenylamino)-4-(trifluoromethyl)-1,6-dihydropyrimidine derivatives
Reactant involved in synthesis of polo-like kinase 1 inhibitors
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Synthesis and SAR of new pyrazolo [4, 3-h] quinazoline-3-carboxamide derivatives as potent and selective MPS1 kinase inhibitors.
Caldarelli M, et al.
Bioorganic & Medicinal Chemistry Letters, 21(15), 4507-4511 (2011)
Synthesis of bactericide Cyprodinil.
Qu J, et al.
Zhejiang Gongye Daxue Xuebao (Journal of Zhejiang University of Technology), 1, 7-7 (2012)
Synthesis of (E)-Methyl 2-(2-((6-cyclopropyl-2-(phenylamino) pyrimidin-4-yloxy) methyl) phenyl)-3-methoxyacrylate and Its Fungicidal Activity.
Li HC, et al.
Agrochemicals, 4, 6-6 (2009)
An unusual Michael addition of 3, 3-dimethoxypropanenitrile to 2-aryl acrylates: a convenient route to 4-unsubstituted 5, 6-dihydropyrido [2, 3-d] pyrimidines.
Berzosa X, et al.
The Journal of Organic Chemistry, 75(2), 487-490 (2009)
Solvent-Free Synthesis of 2-(Phenylamino)-4-(trifluoromethyl)-1, 6-dihydropyrimidine Derivatives Catalyzed by Sulfamic Acid.
Shen S, et al.
Journal of Heterocyclic Chemistry, 49(5), 1033-1037 (2012)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service