8.52373
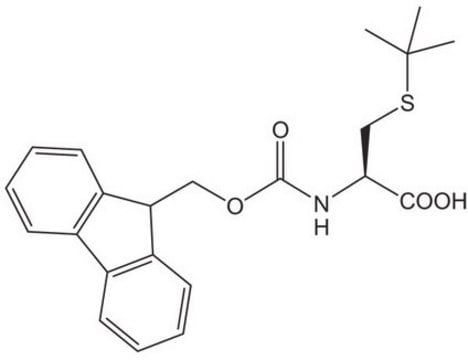
Fmoc-Cys(STmp)-OH
for peptide synthesis, Novabiochem®
Sinónimos:
Fmoc-Cys(STmp)-OH, N-α-Fmoc-S-2,4,6-trimethoxyphenylthio-L-cysteine
About This Item
Productos recomendados
Nombre del producto
Fmoc-Cys(STmp)-OH, Novabiochem®
Nivel de calidad
Línea del producto
Novabiochem®
Formulario
powder
idoneidad de la reacción
reaction type: Fmoc solid-phase peptide synthesis
fabricante / nombre comercial
Novabiochem®
aplicaciones
peptide synthesis
grupo funcional
thiol
temp. de almacenamiento
15-25°C
cadena SMILES
S(Sc4c(cc(cc4OC)OC)OC)C[C@H](NC(=O)OCC1c2c(cccc2)c3c1cccc3)C(=O)O
InChI
1S/C27H27NO7S2/c1-32-16-12-23(33-2)25(24(13-16)34-3)37-36-15-22(26(29)30)28-27(31)35-14-21-19-10-6-4-8-17(19)18-9-5-7-11-20(18)21/h4-13,21-22H,14-15H2,1-3H3,(H,28,31)(H,29,30)/t22-/m0/s1
Clave InChI
GADCBXMSWHDNAU-QFIPXVFZSA-N
Descripción general
Associated Protocols and Technical Articles
Cleavage and Deprotection Protocols for Fmoc SPPS
Fmoc SPPS of Cysteine-Containing Peptides
Literature references:
[1] T. M. Postma, et al. (2012) Org. Lett., 14, 5468.
[2] T. M. Postma & F. Albericio (2013) Org. Lett., 15, 616.
Aplicación
- Synthesis of insulin analogs by regiospecific disulfide bond formation.
- A review on step-wise introduction of disulfide bonds.
- Synthesis of human insulin-like peptide 6.
Nota de análisis
Appearance of substance (visual): powder
Identity (IR): passes test
Enantiomeric purity: ≥ 99.5 % (a/a)
Assay (HPLC, area%): ≥ 94.0 % (a/a)
Purity (TLC(011A)): ≥ 98 %
Solubility (1 mmole in 2 ml DMF): clearly soluble
Ethyl acetate (HS-GC): ≤ 0.5 %
Acetate (IC): ≤ 0.05 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
Información legal
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 1
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Novabiochem® offers orthogonally protected amino acids for peptide synthesis, including cyclic and branched peptides.
Novabiochem® offers orthogonally protected amino acids for peptide synthesis, including cyclic and branched peptides.
Novabiochem® offers orthogonally protected amino acids for peptide synthesis, including cyclic and branched peptides.
Novabiochem® offers orthogonally protected amino acids for peptide synthesis, including cyclic and branched peptides.
Protocolos
Overcome challenges in synthesis and disulfide bond formation with protocols for Fmoc solid-phase peptide synthesis of peptides with cysteine and methionine.
Overcome challenges in synthesis and disulfide bond formation with protocols for Fmoc solid-phase peptide synthesis of peptides with cysteine and methionine.
Overcome challenges in synthesis and disulfide bond formation with protocols for Fmoc solid-phase peptide synthesis of peptides with cysteine and methionine.
Overcome challenges in synthesis and disulfide bond formation with protocols for Fmoc solid-phase peptide synthesis of peptides with cysteine and methionine.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico