380563
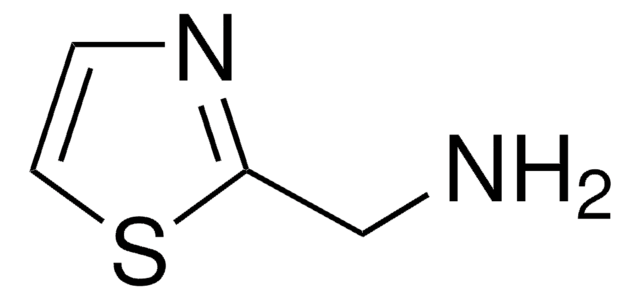
2-Amino-5-methylthiazole
98%
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C4H6N2S
Número de CAS:
Peso molecular:
114.17
Beilstein:
109603
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Ensayo
98%
Formulario
powder
mp
93-98 °C (lit.)
cadena SMILES
Cc1cnc(N)s1
InChI
1S/C4H6N2S/c1-3-2-6-4(5)7-3/h2H,1H3,(H2,5,6)
Clave InChI
GUABFMPMKJGSBQ-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
2-Amino-5-methylthiazole is a heterocyclic building block. It is one of the major alkaline metabolite of tenoxicam (TX) and meloxicam (MX).
Aplicación
2-Amino-5-methylthiazole may be used in the preparation of acrylamide monomer, 5-methyl-2-thiozyl methacrylamide (MTMAAm).
2-Amino-5-methylthiazole may be used in the preparation of mixed-ligand dien-Cu(II) complexes (dien=diethylenetriamine). It may be used in the preparation of a new series of Schiff mono/dibase coordination compounds, having potential anticancer and antibacterial activities.
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - STOT RE 2
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Riccardo Baron et al.
Chemphyschem : a European journal of chemical physics and physical chemistry, 9(7), 983-988 (2008-04-18)
The binding of 2-amino-5-methylthiazole to the W191G cavity mutant of cytochrome c peroxidase is an ideal test case to investigate the entropic contribution to the binding free energy due to changes in receptor flexibility. The dynamic and thermodynamic role of
A Th Chaviara et al.
Journal of inorganic biochemistry, 99(2), 467-476 (2004-12-29)
Two novel mononuclear Cu(II) coordination compounds of the type [Cu(dptaS)Cl(2)] and [Cu(dptaS)Br(2)] (dptaS=1,3-propanediamine, N(1)-[3-aminopropyl]-N(3)-[2-thienylmethylidene] or Schiff mono-base of dipropylenetriamine with 2-thiophene-carboxaldehyde) were prepared by the hydrolysis of the di-bases, [Cu(dptaSS)Cl(2)] and [Cu(dptaSS)Br(2)] (dptaSS=1,3-propanediamine, N(1)-[2-thienylmethylidene]-N(3)-[[2-thienylmethylidene]aminopropyl] or Schiff di-base of dipropylenetriamine with
A Th Chaviara et al.
Journal of inorganic biochemistry, 98(8), 1271-1283 (2004-07-24)
A new series of coordination compounds of the starting materials [Cu(dienX(2)Y(2))] and their adducts [Cu(dienXXY(2))(2a-5mt)] (where dien=diethylenetriamine, dienXX=Schiff bases of diethylenetriamine with 2-furaldehyde or 2-thiophene-carboxaldehyde, X=O, S, Y=Cl, Br, NO(3) and 2a-5mt=2-amino-5-methylthiazole) were synthesized by stepwise reactions and their structures
C A Bolos et al.
Journal of inorganic biochemistry, 88(1), 25-36 (2001-12-26)
The reaction of [Cu(dien)NO(3)]NO(3) with 2-amino-5-methylthiazole (2A5MT), 2-amino-2-thiazoline (2A-2Tzn), imidazole (im), N,N'-thiocarbonyldiimidazole (Tcdim), 2-aminothiazole (2AT) and 2-ethylimidazole (2Etim), gave a new series of mixed-ligand compounds of the general formula [Cu(dien)(B)NO(3))]NO(3); (dien, diethylenetriamine; B, 2A5MT, 2A-2Tzn, im, Tcdim, 2AT and 2Etim).
Synthesis and characterization of new chelating resin: adsorption study of copper (II) and chromium (III) ions.
Sacmaci S, et al.
Journal of Macromolecular Science, Part A: Pure and Applied Chemistry, 47(6), 552-557 (2010)
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico