711144
Potassium 1H-pyrazole-3-trifluoroborate
Synonym(s):
Potassium 1H-pyrazole-5-trifluoroborate, Potassium pyrazole-5-trifluoroborate
About This Item
Recommended Products
form
powder
Quality Level
mp
196-200 °C
SMILES string
[K+].F[B-](F)(F)c1cc[nH]n1
InChI
1S/C3H3BF3N2.K/c5-4(6,7)3-1-2-8-9-3;/h1-2H,(H,8,9);/q-1;+1
InChI key
QNHYBMZHGRWURK-UHFFFAOYSA-N
Application
Organotrifluoroborates as versatile and stable boronic acid surrogates.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Bench-stable Potassium Organotrifluoroborates enable diverse C-C bond formation reactions.
Bench-stable Potassium Organotrifluoroborates enable diverse C-C bond formation reactions.
Bench-stable Potassium Organotrifluoroborates enable diverse C-C bond formation reactions.
Bench-stable Potassium Organotrifluoroborates enable diverse C-C bond formation reactions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
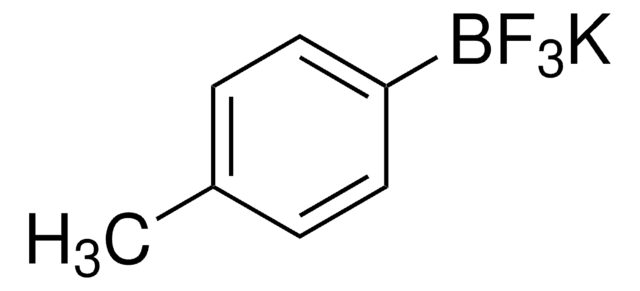
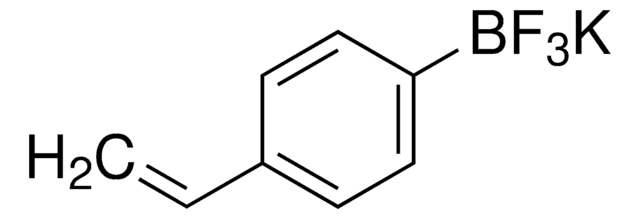
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)

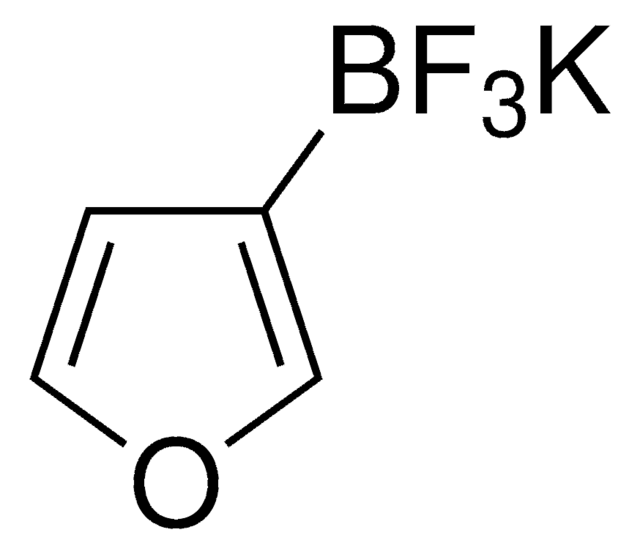


![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II), complex with dichloromethane](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)

