800814P
Avanti
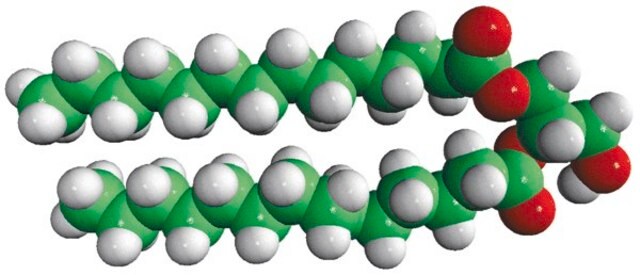
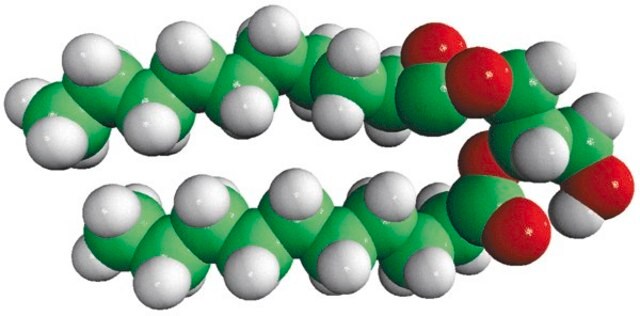
14:0 DG
1,2-dimyristoyl-sn-glycerol, powder
Synonym(s):
1,2-ditetradecanoyl-sn-glycerol; DG(14:0/14:0/0:0)
About This Item
Recommended Products
Assay
>99% (TLC)
form
powder
packaging
pkg of 1 × 10 mg (800814P-10mg)
pkg of 1 × 25 mg (800814P-25mg)
manufacturer/tradename
Avanti Research™ - A Croda Brand 800814P
lipid type
neutral glycerides
neutral lipids
shipped in
dry ice
storage temp.
−20°C
InChI
1S/C31H60O5/c1-3-5-7-9-11-13-15-17-19-21-23-25-30(33)35-28-29(27-32)36-31(34)26-24-22-20-18-16-14-12-10-8-6-4-2/h29,32H,3-28H2,1-2H3/t29-/m0/s1
InChI key
JFBCSFJKETUREV-LJAQVGFWSA-N
1 of 4
This Item | 800810P | 840445P | 800820P |
|---|---|---|---|
| form powder | form powder | form powder | form powder |
| manufacturer/tradename Avanti Research™ - A Croda Brand 800814P | manufacturer/tradename Avanti Research™ - A Croda Brand 800810P | manufacturer/tradename Avanti Research™ - A Croda Brand | manufacturer/tradename Avanti Research™ - A Croda Brand 800820P |
| shipped in dry ice | shipped in dry ice | shipped in dry ice | shipped in dry ice |
| packaging pkg of 1 × 10 mg (800814P-10mg), pkg of 1 × 25 mg (800814P-25mg) | packaging pkg of 1 × 10 mg (with screw cap (800810P-10mg)) | packaging pkg of 1 × 1 g (840445P-1g), pkg of 1 × 200 mg (840445P-200mg), pkg of 1 × 25 mg (840445P-25mg), pkg of 1 × 500 mg (840445P-500mg) | packaging pkg of 1 × 5 mg (800820P-5mg) |
| storage temp. −20°C | storage temp. −20°C | storage temp. −20°C | storage temp. −20°C |
General description
Diacylglycerol mimicks the effects of the tumor-promoting compounds phorbol esters.
Application
Biochem/physiol Actions
Packaging
Storage and Stability
Other Notes
Dry samples of diacylglycerol in chloroform, using a stream of nitrogen. Dissolve the residue in an appropriate volume of ethanol or DMSO, then dilute to the desired aqueous medium.
Most biological responses saturate at 20 to 250 μM sn-1,2-dioctanoylglycerol. Only sn-1,2 isomers appear to be active.
Legal Information
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![23:2 Diyne PE [DC(8,9)PE] 1,2-bis(10,12-tricosadiynoyl)-sn-glycero-3-phosphoethanolamine, powder](/deepweb/assets/sigmaaldrich/product/images/228/422/4e95f75c-14fa-4117-a383-2eff73fa927f/640/4e95f75c-14fa-4117-a383-2eff73fa927f.jpg)