P56100
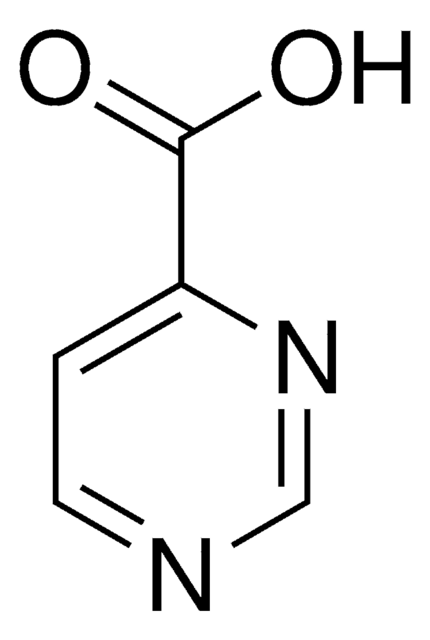
Pyrazinecarboxylic acid
99%
Synonym(s):
Pyrazinoic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C5H4N2O2
CAS Number:
Molecular Weight:
124.10
Beilstein:
112305
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
powder
mp
222-225 °C (dec.) (lit.)
SMILES string
OC(=O)c1cnccn1
InChI
1S/C5H4N2O2/c8-5(9)4-3-6-1-2-7-4/h1-3H,(H,8,9)
InChI key
NIPZZXUFJPQHNH-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Mohamed Abdel-Aziz et al.
European journal of medicinal chemistry, 45(8), 3384-3388 (2010-05-22)
A series of pyrazine-2-carboxylic acid hydrazide derivatives were synthesized and screened for their activity against Mycobacterium tuberculosis. The results show that pyrazine-2-carboxylic acid hydrazide-hydrazone derivatives 3a-l were less active than pyrazinamide. In contrast, the N(4)-ethyl-N(1)-pyrazinoyl-thiosemicarbazide 4 showed the highest activity
Anthony D Baughn et al.
Antimicrobial agents and chemotherapy, 54(12), 5323-5328 (2010-09-30)
The pyrazinamide (PZA) analog 5-chloropyrazinamide (5-Cl PZA) is active against mycobacterial species, including PZA-resistant strains of Mycobacterium tuberculosis. In M. smegmatis, overexpression of the type 1 fatty acid synthase (FAS I) confers resistance to 5-Cl PZA, a potent FAS I
Wanliang Shi et al.
Science (New York, N.Y.), 333(6049), 1630-1632 (2011-08-13)
Pyrazinamide (PZA) is a first-line tuberculosis drug that plays a unique role in shortening the duration of tuberculosis chemotherapy. PZA is hydrolyzed intracellularly to pyrazinoic acid (POA) by pyrazinamidase (PZase, encoded by pncA), an enzyme frequently lost in PZA-resistant strains
Takashi Iwanaga et al.
The Journal of pharmacology and experimental therapeutics, 320(1), 211-217 (2006-10-18)
Serum uric acid (SUA) is currently recognized as a risk factor for cardiovascular disease. It has been reported that an angiotensin II receptor blocker (ARB), losartan, decreases SUA level, whereas other ARBs, such as candesartan, have no lowering effect. Because
H J Kim et al.
The international journal of tuberculosis and lung disease : the official journal of the International Union against Tuberculosis and Lung Disease, 16(1), 98-103 (2012-01-13)
Pyrazinamide (PZA), one of the most effective anti-tuberculosis drugs, becomes toxic to Mycobacterium tuberculosis when converted to pyrazinoic acid by pyrazinamidase (PZase). PZA resistance is caused mainly by the loss of enzyme activity by mutation. To investigate the patterns of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service